Quick Answer: No, boiling water is not the same as distilled water.
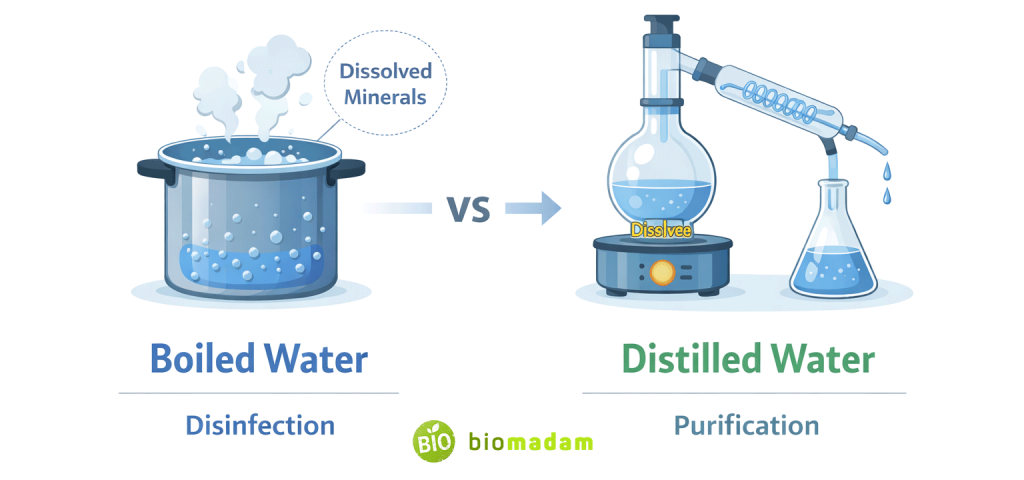
Boiling water kills most disease-causing microorganisms but does not remove dissolved minerals, salts, or chemical contaminants. Distilled water is produced by boiling water and then condensing the steam, which removes most dissolved solids and impurities, resulting in highly purified water.
The key difference:
- Boiling disinfects water.
- Distillation purifies water.
According to public health guidelines (such as CDC and WHO recommendations), boiling water for at least 1 minute is sufficient to inactivate most disease-causing microorganisms under normal conditions.
This article explains the difference between boiled water and distilled water from chemistry perspective, including how each process works, what impurities are removed, and which option is suitable for drinking, laboratory use, and household applications.
Boiling Water and Distilled Water Comparison
| Feature | Boiled Water | Distilled Water |
| Purpose | Disinfection | Purification |
| Removes Bacteria | Yes (most pathogens) | Yes |
| Removes Dissolved Minerals | No | Yes (mostly) |
| Removes Chemical Contaminants | No | Removes most non-volatile contaminants; some volatile compounds may remain. |
| Process | Heating to 100°C | Boiling + Condensation |
| Taste | Normal / slightly mineral | Flat (no minerals) |
| Used In | Drinking (emergency), cleaning | Laboratories, medical use, batteries |
Scientifically, boiling changes water from liquid to vapor, but unless the vapor is collected separately, dissolved solids remain in the original water, whereas distillation separates water molecules from impurities through phase change followed by condensation into a separate container.
Core Concept: Disinfection vs Purification
The confusion exists because both processes involve boiling water.
However:
- Boiling → kills microorganisms
- Distillation → removes dissolved substances through phase change
When water boils, it changes from liquid to vapor. In distillation, that vapor is collected and condensed separately, leaving most impurities behind.
What is Boiled Water?
Boiled water is water heated to its boiling point (100°C at sea level).
At higher altitudes, water boils at lower temperatures due to reduced atmospheric pressure. Therefore, health authorities recommend boiling water for at least 3 minutes in high-altitude areas to ensure proper disinfection.
Boiling:
- Kills most bacteria, viruses, and parasites
- Does NOT remove dissolved salts, heavy metals, or chemical pollutants
- Is recommended during water contamination emergencies
Important Scientific Clarification
- Boiling does not make water chemically pure. Boiling is a physical change involving phase transition, as explained in the Difference Between Physical And Chemical Change.
- It only disinfects by destroying pathogens through heat.
What is Distilled Water?

Distilled water is produced by:
- Heating water to produce steam
- Separating the steam from impurities
- Condensing the steam into liquid form
Because dissolved salts and most impurities do not evaporate with water vapor, they remain behind in the boiling chamber.
Scientific Note
Distillation removes:
- Minerals
- Salts
- Many microorganisms
- Most dissolved solids
Distillation significantly reduces Total Dissolved Solids (TDS), which measure the concentration of dissolved minerals and salts in water.
However, some volatile substances (like certain organic compounds) may still carry over unless additional filtration is used.
Step-By-Step Process Comparison
Boiling Process
- Heat water to 100°C
- Water forms bubbles
- Microorganisms are inactivated
No separation occurs — impurities remain in the water.
Distillation Process
- Heat water to boiling
- Steam rises
- Steam cools and condenses
- Condensed water collected separately
Impurities remain in the original container.
Key Scientific Differences
Removal of Microorganisms
Both boiling and distillation eliminate most pathogens.
Removal of Minerals
- Boiling → minerals remain
- Distillation → minerals removed
Removal of Chemical Contaminants
- Boiling → ineffective
- Distillation → removes most non-volatile contaminants
Chemical Purity
- Boiled water = disinfected
- Distilled water = highly purified
The classification of distilled water as a pure substance is discussed in the Difference Between Pure Substance and Mixture.
Is Distilled Water Safe to Drink?
Distilled water is safe to drink occasionally.
However:
- It contains no minerals, unlike hard water discussed in the Difference Between Hard Water and Soft Water.
- Long-term exclusive consumption is not recommended unless dietary minerals are adequate
In normal conditions, mineral-containing drinking water is preferred.
Applications of Boiled Water
- Emergency drinking water treatment
- Cooking
- Cleaning utensils
- Disinfection during contamination events
Applications of Distilled Water
- Laboratory experiments
- Medical equipment
- Pharmaceutical manufacturing
- Car batteries
- Steam irons
- Aquariums (controlled mineral environments)
Distilled water is preferred where mineral interference must be avoided.
Because distillation requires sustained heating and condensation, it consumes more energy than simple boiling and is therefore less commonly used for large-scale municipal water treatment.
Which is Better: Boiled or Distilled Water?

It depends on the purpose:
- For emergency drinking water → Boiled water is sufficient.
- For laboratory or medical use → Distilled water is required.
- For appliances like steam irons or car batteries → Distilled water is preferred to prevent mineral buildup.
- For daily hydration → Mineral-containing drinking water is generally preferred for taste and balanced mineral intake.
Common Misconception
Boiling water does not make it chemically pure.
It only destroys pathogens. Dissolved minerals and heavy metals remain.
Distillation involves both boiling and condensation, which physically separates water from most impurities.
One-Line Summary
- Boiling disinfects water.
- Distillation purifies water.
FAQs
Does boiling water make it distilled?
No. Distilled water requires condensation of steam into a separate container.
Does boiling remove heavy metals?
No. Heavy metals remain in boiled water.
Why does distilled water taste flat?
Because it lacks dissolved minerals that give natural water its taste. The relationship between distilled water and water hardness is further clarified in Is Distilled Water Soft Water?
Is distilled water the purest form of water?
Distilled water is purified through phase separation, removing most dissolved solids and microorganisms. Laboratory-grade deionized water can be even more chemically refined.
Can I use boiled water instead of distilled water?
- For drinking during emergencies, yes.
- For laboratory or technical use, no — distilled water is required.
The Bottom Line
Boiled water and distilled water are not the same.
Boiling disinfects water by killing microorganisms but leaves dissolved minerals and chemical contaminants behind. Distillation purifies water by separating it from most impurities through evaporation and condensation.
Understanding this difference is essential for safe drinking, laboratory accuracy, and industrial applications.

The Science Editorial Team creates clear, accurate, and student-friendly explanations of scientific concepts across biology, chemistry, physics, taxonomy, anatomy, and related life sciences. Content is written for educational and informational purposes using standard academic references and reliable sources. Read more about the → Science Editorial Team
