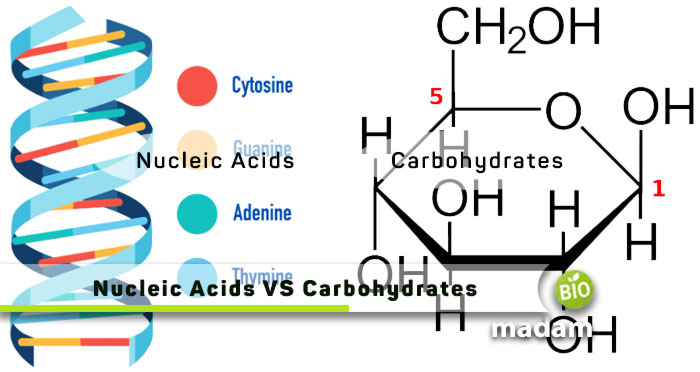
Our body requires biomolecules, and macro and micronutrients to grow and replicate. Carbohydrates and nucleic acids are such important components contributing to different processes within the body. However, they are quite distinct in structure and function. Carbohydrates are one of the most important macronutrients in the body that provide energy. At the same time, a nucleic acid is a critical component of DNA and RNA. Keep reading to know all the differences between nucleic acids and carbohydrates.
Comparison Table
| Factors | Nucleic Acids | Carbohydrates |
| Definition | Genetic information storing and expression compounds | Organic molecules provide energy to the cells |
| Composition | Pentose sugar, nitrogen base, and phosphate group | Hydrogen, oxygen, and carbon atoms |
| Basic Unit | Nucleotides | Monosaccharides |
| Linkages | Phosphodiester | Glycosidic |
| Types | RNA & DNA | Monosaccharides, disaccharides, oligosaccharides, & polysaccharides |
| Storage | Nucleus | Liver and muscle tissues |
| Examples | RNA, DNA | Fructose, maltose, sucrose |
What are Nucleic Acids?
Nucleic acids are important organic molecules in the body that make up genetic material. They provide structure to hereditary molecules, including RNA, DNA, genes, etc. Nucleic acids comprise biopolymers composed of repeated sets of monomers forming nucleotides. The nucleotides eventually form nucleic acids.
Here is the structure of nucleic acids thoroughly for better understanding.
Structure of Nucleic Acid
Nucleic acid comprises repeating nucleotide monomers acting as the basic structural unit. Understanding the structure of nucleotides will help comprehend the structure of nucleic acid. The three main parts of nucleotides include a nitrogen base, pentose sugar, and phosphate group.
Nitrogen Base
As the name indicates, the nitrogen base is the foundation of each nucleotide. It binds to hydrogen, carbon, and oxygen to form a ring structure. The ring structure could be pyrimidine or purine depending on the composition. Pyrimidine ring structures have single rings, whereas double ring nitrogen bases are known as purines.
Pyrimidines are composed of cytosine, thymine, and uracil. Alternatively, purines contain guanine and adenine and are larger than pyrimidines.
Pentose Sugar
A 5-sugar structure, also known as pentose sugar, is another important component of nucleotides. Thus, we can say that nucleic acid contains carbohydrates. Nucleic acids may have ribose or deoxyribose sugars giving rise to distinct RNA and DNA structures. Both nucleic acids contain five carbon atoms and one oxygen atom. Furthermore, the carbon atoms are attached to hydrogen atoms and hydroxyl groups.
Phosphate Group
The phosphate group is a phosphorus atom having four negatively charged oxygen atoms attached.
Function of Nucleic Acid
Nucleic acids are majorly involved in the expression and storage of hereditary information. RNA and DNA contribute to the production of new components. DNA carries genomic information from the parent cell to the daughter cells.
Types of Nucleic Acid
Nucleic acids are of two types: Ribonucleic acid (RNA) and Deoxyribonucleic Acid (DNA).
RNA
RNA is critical for producing different types of proteins as ribosomes translate the data into proteins. RNA is involved in the process of transcription and carrying out protein biosynthesis.
RNA contains a ribose sugar in which the hydroxyl groups are joined to the second and third carbon. They are single-stranded and contain uracil paired with adenine.
DNA
DNA is the main part of genes and alleles for storing and translating genetic information. The nucleotides bind to each other in a specific manner. Adenine binds with thymine, while guanine binds to cytosine. The DNA contains all the information for the production of proteins as well.
The deoxyribose sugar in DNA has a hydroxyl group joined to the third carbon atom. The second carbon atom contains a hydrogen atom.
What are Carbohydrates?
Besides fats and proteins, carbohydrates are important macronutrients. Carbohydrates are the energy source required in various cellular functions including nerve transmissions in sympathetic and parasympathetic nervous system.
They are the most abundant organic compounds found in multiple foods around us. Plants produce carbohydrates through photosynthesis, making 60 – 90% of the dry matter. Animals obtain dietary carbohydrates from plants for normal body function.
Natural carbohydrates include sugars, starches, and fiber; they are together known as total carbohydrates.

Structure of Carbohydrates
Carbohydrates are organic molecules containing carbon, oxygen, and hydrogen. The basic chemical formula for carbohydrates is C6H12O6. The carbs having this structure are known as monosaccharides which combine to form further structures. They contain a chain of carbons, a hydroxyl group, and an aldehyde or ketone.
Function of Carbohydrates
Carbohydrates are essential for providing energy in animals’ and plants’ life stages. Organisms use different forms of carbohydrates for growth and development. The brain requires energy supplied by carbohydrates to transmit signals. However, it does not obtain energy from saturated or unsaturated fats. Your body metabolizes carbohydrates into glucose required by body cells, tissues, and organs.
Maintaining a steady supply of glucose is important, or you may experience hypoglycemia or hyperglycemia. The increased amount of glucose in the blood may give rise to diabetes whereas hypoglycemia results from low blood sugar.
Let’s tell you about the types of carbohydrates in detail.
Types of Carbohydrates
Carbohydrates are majorly divided into four groups according to their structure. The four main classes of carbohydrates are monosaccharides, disaccharides, oligosaccharides, and polysaccharides.
Monosaccharides
Monosaccharides are the most simplified form of carbohydrates that cannot be hydrolyzed. They may further be named triose, tetrose, pentose, and hexose, depending on the number of carbon atoms in each molecule.
- Triose – 3 Carbons
- Tetrose – 4 Carbons
- Pentose – 5 Carbons
- Hexose – 6 Carbons
Glucose, ribose, and sucrose are examples of monosaccharides.
Disaccharides
The word disaccharide comprises “Di + Saccharide”, translating to two carbohydrates. Disaccharides comprise two monosaccharide molecules joined together with a glycosidic bond. Monosaccharides may form alpha or beta bonds. Alpha bonds form when the OH group on Carbon 1 of the first glucose lies below the ring plain. On the other hand, the OH group placement on carbon 1 above the ring plain results in a beta bond.
Maltose and lactose are disaccharide examples.
Oligosaccharides
Oligosaccharides are the intermediate of disaccharides and polysaccharides. They form by the bonding of three or more monosaccharides. Oligosaccharides have been seen to improve hindgut health by facilitating prebiotic and probiotic growth.
Stachyose and Raffinose are oligosaccharides composed of glucose, galactose, and fructose.
Polysaccharides
Polysaccharides are complex carbohydrates produced by the combination of multiple polymers of monosaccharides. They act as energy-storage units in plants and animals and offer structural support. The polysaccharide chain may be branched or unbranched, containing one or more types of monosaccharides. The weight of polysaccharides is 100,000 daltons or above, depending on the number of monosaccharide molecules.
Cellulose, starch (amylose and amylopectin), and glycogen are polysaccharides.
Similarities Between Nucleic Acid and Carbohydrates
- Both of them contain carbon atoms important to living organisms.
- Nucleic acids and carbohydrates comprise monosaccharides.
- Both are produced by living organisms.
Differences Between Nucleic Acid and Carbohydrates
Definition
Nucleic Acids
Nucleic acids are large biomolecules that carry protein synthesis data and genetic information.
Carbohydrates
Carbohydrates are organic molecules essential for growth and development in all organisms.
Composition
Nucleic Acids
Nucleic acids comprise a pentose sugar, nitrogen base, and phosphate group.
Carbohydrates
Carbohydrates are composed of hydrogen, oxygen, and carbon atoms forming macromolecules.
Basic Unit
Nucleic Acids
Nucleic acids contain nucleotides as the basic building unit.
Carbohydrates
Monosaccharides are the basic components of carbohydrates.
Linkages
Nucleic Acids
A phosphodiester bond facilitates the formation of nucleic acids through nucleotide formation.
Carbohydrates
Carbohydrates contain glycosidic bonds that bind monomers to produce other structures.
Types
Nucleic Acids
Nucleic acids are of two types – Ribonucleic acid (RNA) and Deoxyribonucleic acid (DNA).
Carbohydrates
Carbohydrates are categorized as monosaccharides, disaccharides, oligosaccharides, and polysaccharides.
Functions
Nucleic Acids
Nucleic acids are involved in storing and expressing hereditary information for protein synthesis and replication.
Carbohydrates
Carbohydrates are the body’s major energy source to carry out cellular and bodily functions.
Storage
Nucleic Acids
Nucleic acids are stored in the nucleus of each cell in the form of DNA or RNA in the synthesis phase.
Carbohydrates
Carbohydrates are converted into glycogen and stored in connective tissues in the liver and in muscles.
Examples
Nucleic Acids
RNA and DNA are the nucleic acids found in living organisms.
Carbohydrates
Sucrose, fructose, maltose, glucose, and lactose are carbohydrate examples.
The Bottom Line
Nucleic acids and carbohydrates are essential for survival and reproduction. Nucleic acids act as the storage center for hereditary information within the nucleus that perform various other functions. Alternatively, carbohydrates provide the body cells and tissues with energy to perform normal bodily functions. They are composed of structural units that form long chains to create different forms. It is important to take carbohydrates in adequate sources to keep your bodily functions running properly.
FAQs
Are nucleic acids considered carbohydrates?
Nucleic acids are not considered carbohydrates, yet they contain a pentose ring as a part of their structure. DNA contains deoxyribose, while RNA has ribose sugar in its structure.
What is the difference between amino acids and carbohydrates?
Amino acids are the body’s building blocks, whereas carbohydrates are macromolecules comprising different types of sugars. Amino acids combine to produce large protein molecules.
Which is more important to living things, nucleic acids or carbohydrates?
Nucleic acids and carbohydrates are essential to life in their own ways. Cells will not be able to grow or replicate in the absence of any of them as the basic structure of nucleotides also contains monosaccharide sugar.

The Biomadam Content Team develops and manages educational and informational articles published on Biomadam. Content is prepared using standard reference materials and follows internal editorial guidelines to ensure clarity and consistency.