Enzymes are biological catalysts essential for sustaining life. Every metabolic process inside a living cell—from digestion and respiration to growth and repair—depends on enzymes. However, cellular reactions cannot run uncontrollably. To maintain balance, organisms rely on regulatory enzymes, which control the rate and direction of biochemical pathways.
This article explains regulatory enzymes, their types, mechanisms of regulation, and biological significance, in a way that supports both conceptual understanding and exam preparation.
What Are Enzymes?
Enzymes are high-molecular-weight biological molecules, mostly proteins, that increase the rate of biochemical reactions without being consumed in the process.
Key Characteristics of Enzymes
- Highly specific to their substrate
- Act as biological catalysts
- Remain chemically unchanged after the reaction
- Mostly protein in nature (exception: ribozymes)
Enzymes are present in all living cells, where they regulate metabolic activities essential for survival.
What Are Regulatory Enzymes?
Regulatory enzymes are enzymes that control the rate of metabolic pathways by responding to cellular signals such as substrate concentration, end products, or signaling molecules.
They usually catalyze:
- Rate-limiting steps
- Committed steps in metabolic pathways
Their activity determines when, where, and how fast a biochemical pathway operates.
Example: The synthesis of hormones, neurotransmitters, and metabolic intermediates occurs through multi-step pathways controlled by regulatory enzymes.
Why Is Enzyme Regulation Necessary?
Cells constantly adjust their metabolism based on:
- energy demand
- nutrient availability
- environmental conditions
- tissue-specific functions
For example:
- Liver cells require different enzymatic activities than nerve cells.
- Digestive enzyme activity increases after meals and decreases during fasting.
Without regulation, metabolic reactions would:
- waste energy
- accumulate toxic intermediates
- disrupt cellular homeostasis
Types of Enzyme Regulation
Enzyme regulation mainly occurs through two mechanisms: allosteric regulation and covalent modification.
| Feature | Allosteric Regulation | Covalent Regulation |
| Binding Site | Allosteric site | Specific amino acid residue |
| Type of Change | Conformational | Chemical modification |
| Reversibility | Usually reversible | Reversible or irreversible |
| Common Modifiers | Activators / Inhibitors | Phosphate, methyl, acetyl groups |
| Speed of Response | Very fast | Fast to moderate |
| Energy Requirement | No direct ATP use | Often requires ATP |
| Example | Feedback inhibition | Phosphorylation |
| Exam Importance | High | Very high |
Regulatory enzymes are classified based on how their activity is controlled. The two major types are:
- Allosteric Regulation
- Covalent Enzyme Regulation
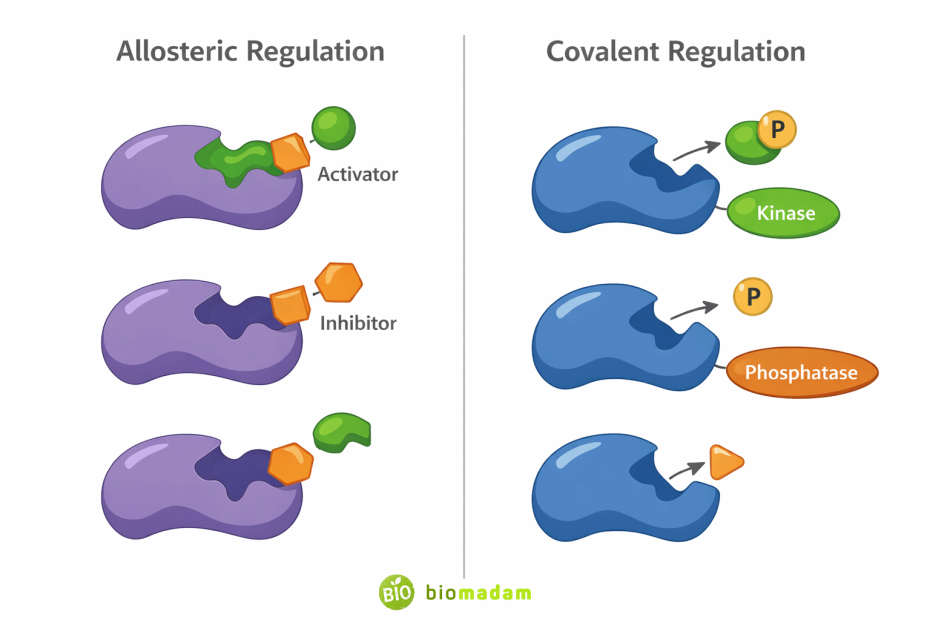
1. Allosteric Regulation

In allosteric regulation, molecules bind to a site on the enzyme other than the active site, called the allosteric site.
Most allosteric enzymes are oligomeric proteins (made of multiple subunits).
Allosteric Activators
- Increase enzyme activity
- Stabilize the active form of the enzyme
- In some cases, the substrate itself acts as an activator (cooperativity)
Allosteric Inhibitors
- Decrease enzyme activity
- Bind at the allosteric site
- May be competitive or non-competitive
Feedback Regulation
- Positive feedback: increases pathway activity
- Negative feedback (feedback inhibition): end product inhibits an early enzyme in the pathway
2. Covalent Enzyme Regulation
Covalent regulation involves chemical modification of the enzyme through the addition or removal of functional groups.
(a) Reversible Covalent Regulation
This type allows enzyme activity to switch on or off reversibly.
Common modifications include:
- Phosphorylation (Serine, Threonine, Tyrosine)
- Methylation
- Acetylation
- Nucleotidylation
Phosphorylation & Dephosphorylation

- Addition of phosphate group → catalyzed by kinases
- Removal of phosphate group → catalyzed by phosphatases
This mechanism plays a central role in:
- signal transduction
- metabolic control
- energy regulation
(b) Irreversible Covalent Regulation
In irreversible regulation:
- Enzymes are activated by proteolytic cleavage
- The process cannot be reversed
Example: Digestive enzymes synthesized as inactive zymogens and activated when needed.
Importance of Regulatory Enzymes
Regulatory enzymes are essential for:
- Maintaining metabolic balance
- Preventing wasteful or harmful reactions
- Coordinating complex biochemical pathways
- Responding to internal and external signals
- Conserving cellular energy
They ensure that metabolic processes occur only when required and at appropriate rates.
Real-Life Applications of Enzyme Regulation

- Regulation of insulin and glucose metabolism
- Drug action targeting regulatory enzymes
- Industrial fermentation control
- Metabolic disorders caused by faulty regulation
- Biotechnology and genetic engineering applications
Summary
- Enzymes = biological catalysts
- Regulatory enzymes control metabolic pathways
- Two main types:
- Allosteric regulation
- Covalent regulation
- Feedback inhibition prevents overproduction
- Phosphorylation is a major regulatory mechanism
Frequently Asked Questions (FAQs)
What are regulatory enzymes?
Regulatory enzymes control the rate of biochemical reactions and metabolic pathways in response to cellular signals.
What is the difference between allosteric and covalent regulation?
Allosteric regulation involves binding at a non-active site, while covalent regulation involves chemical modification of the enzyme.
Why is feedback inhibition important?
It prevents excessive accumulation of end products and maintains metabolic balance.
Are all enzymes regulatory enzymes?
No. Only specific enzymes involved in rate-limiting or control steps act as regulatory enzymes.
What is the most common type of enzyme regulation?
Allosteric regulation is the most common and fastest mechanism used by cells to control metabolic pathways.
MCQs for Practice
1. Which enzymes control the rate of metabolic pathways?
A. Digestive enzymes
B. Structural enzymes
C. Regulatory enzymes ✅
D. Ribozymes
2. Regulatory enzymes usually catalyze which step of a pathway?
A. Random steps
B. Terminal steps
C. Rate-limiting or committed steps ✅
D. Reversible steps only
3. In allosteric regulation, the regulator binds to:
A. Active site
B. Substrate
C. Allosteric site ✅
D. Cofactor
4. Which of the following is an example of negative feedback?
A. Product activates enzyme
B. Substrate increases activity
C. End product inhibits early enzyme ✅
D. Enzyme activates another enzyme
5. Which modification is reversible?
A. Proteolytic cleavage
B. Zymogen activation
C. Phosphorylation ✅
D. Peptide bond cleavage
6. Kinase enzymes are involved in:
A. Removing phosphate groups
B. Adding phosphate groups ✅
C. Breaking peptide bonds
D. Binding substrates
7. Irreversible enzyme regulation commonly involves:
A. Allosteric inhibitors
B. Phosphatases
C. Proteolytic cleavage ✅
D. Cooperativity
8. Most allosteric enzymes are:
A. Monomeric
B. Lipid-based
C. Oligomeric proteins ✅
D. RNA molecules

The Science Editorial Team creates clear, accurate, and student-friendly explanations of scientific concepts across biology, chemistry, physics, taxonomy, anatomy, and related life sciences. Content is written for educational and informational purposes using standard academic references and reliable sources. Read more about the → Science Editorial Team