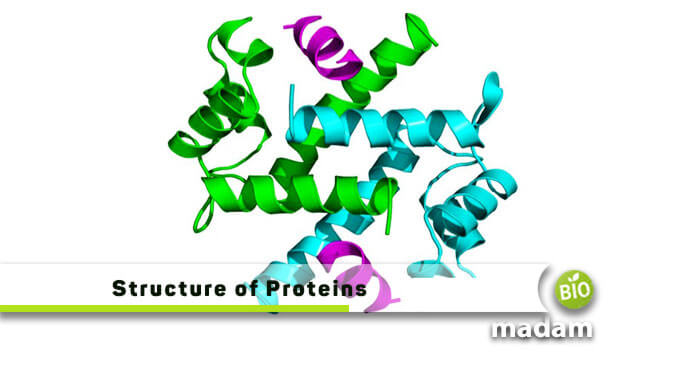
The macromolecules occurring in ample amounts in all the cells are called proteins. These units are the most important for living beings. Hundreds and thousands of protein molecules of different sizes constitute a living body.
Proteins appear when two amino acids link together through a peptide bond. They are involved in many chemical processes and attain elevated nutritional value.
In 1838 Jons Jacob introduced the term “Proteins,” derived from the Greek word “Proteios.” These are organ-specific from species to species. Approximately 20 different amino acids naturally occur in nature.
Proteins with the same function will have the same structure as well. The human body contains 10000 different kinds of proteins performing various functions. You can even find their sequence using Expasy Translate Tool.
A plant can synthesize amino acids, but animals cannot. You will comprehend the structure, properties, and sources of proteins after going through this article.
Peptide and Proteins
The compound formed by condensation of two or more similar or different amino acids is called peptides. They contain –CO-NH- bond in their structure.
It is called peptide bond or peptide linkage. The bond formed between the carbonyl group of one amino acid and an amino group of another amino acid with the elimination of a water molecule is called a peptide bond.
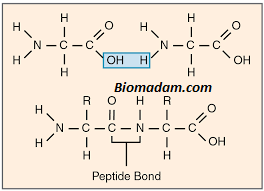
Two amino acids join to form three dipeptide units that further generate a tripeptide. A large number of amino acids join by peptide bonds and make a polypeptide. A polypeptide is a peptide with 10,000 molecular weight. Whereas a Peptide with a molecular mass of more than 10,000 is called protein. The human body contains at least 10,000 different proteins. They are present in the skin, hair, muscles, and non-bony tissues of the body. Let’s check out the structure and properties of polypeptides and proteins below.
Structure and Properties
According to Biochemistry study, the amino acid composition of protein consists of a long chain of alpha-amino acids. α- amino acids define a unit having an amino group (−NH2), a carboxyl group, and an alkyl group
Acid nature of Amino Acid
In the acidic condition, pH is less than 4, and the –COO group combines with a hydrogen atom and converts it into the –COOH group, which is uncharged.
Alkali Nature of Amino Acid
In alkali, condition pH remains above 9, the amino group (−NH+3) releases hydrogen ion and converts it into (−NH2).
What is Zwitter Ion?
The molecule of amino acid consisting of dipolar ions is called a Zwitter ion. These amino acid carries both positive and negative charge and do not migrate with an electric field.
More than 100 amino acids are present, but almost 20 of them occur naturally.
Formation of Protein Molecules
Different alpha-amino acids link together through a peptide bond to form a long-chain molecule called protein.
What is Peptide Bond?
A peptide bond is formed between the amino and carboxyl groups of two different amino acids through a peptide linkage. When this peptide molecule reacts with another amino acid to eliminate water, it yields a tripeptide.
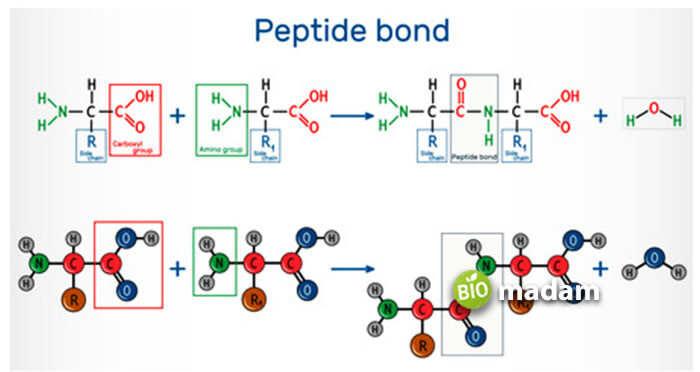
We generally discuss the N- terminal or free alpha-amino acid on the left side and the C-terminal or free carboxyl group found on the right side. These macromolecules are joined through a peptide bond. Many amino acids are linked and form the structure of the protein.
A long peptide chain involves the linkage of more than 100 amino acids.
Classification of Proteins

Proteins have three classes based on physicochemical properties
- Simple Proteins
- Compound Proteins
- Derived Proteins
Simple Proteins
Proteins yield amino acids on hydrolysis, where their derivatives are called simple proteins. Examples include collagen, globulins, legumin, albumins, etc. Globulins are globular proteins in animals that are insoluble in water but soluble in a dilute salt solution. Similarly, body muscles contain lactoglobulins. Legumin and collagen are present in the connective tissues of the body. They are abundantly present, forming 25 to 35% of body protein. They usually either comprise fibrous or globular proteins.
Compound or Conjugate proteins
These molecules are attached to some non-protein groups called conjugated proteins, such as phospho-proteins conjugate with phosphoric acid and lipoproteins with lipids. (Cholesterol, Lecithin)
Derived Proteins
These are procured from simple and conjugated proteins that can be either primary or secondary derived proteins. Some examples include metaproteins and milk from the primary section, while proteases, peptides, peptones, etc., are from the secondary.
Protein Sources

There are several protein sources, including meat, vegetables, poultry, seafood, soy products, seed, nuts, eggs, etc. To gain more protein in the morning, you can store eggs to cook them instantly.
Alternative Sources
Numerous alternative sources like whey and soy hump are utilized for proteins. Its benefits mostly meet the person who exercises regularly because whey proteins are better for regenerating muscles & mass.
What is Whey Protein?

Whey protein is a by-product of cheese making, basically, the watery portion that separates. This protein is usually utilized to improve athletic performance and strengthens the muscles. People with HIV defects consume whey protein which helps them reduce their weight loss problem. Moreover, it helps prevent allergies, infections and certain diseases in infants.
Final Statement
So, proteins are complex macromolecules composed of amino acid chains that perform many critical functions in the body. From structural proteins that provide physical support to functional proteins involved in metabolism, cell signaling, and immune response, proteins enable life as we know it. While many dietary sources provide protein, certain populations like athletes may benefit from supplemental forms like whey. As researchers continue to uncover new information about protein structure and function, innovative medical and dietary applications will likely arise. Understanding the diversity of proteins and their sources is key to promoting optimal health and biological functioning.

The Biomadam Content Team develops and manages educational and informational articles published on Biomadam. Content is prepared using standard reference materials and follows internal editorial guidelines to ensure clarity and consistency.