Quick Answer: Organic compounds are carbon-based molecules that form the foundation of life and modern civilization.
They build cells, store energy, carry genetic information, power fuels, and create medicines, plastics, and countless industrial products.
Without organic compounds, life and modern industry would not exist.
In simple terms, organic compounds are important because they form the chemical basis of life and modern materials.
What This Article Covers
- What organic compounds are
- Why carbon is unique
- Role in living organisms
- Importance in agriculture and medicine
- Industrial and fuel applications
- Environmental relevance
- Common misconceptions
- Practical significance
Core Concept: What Are Organic Compounds?
Organic compounds are primarily carbon-containing compounds characterized by carbon–hydrogen (C–H) bonds, typically bonded to hydrogen, oxygen, nitrogen, sulfur, or phosphorus.
They are a specific type of compound formed by bonded atoms and differ from the broader concepts of molecules and compounds studied in basic chemistry.
The defining feature is the carbon backbone.
Carbon forms strong covalent bonds. It can create chains, rings, and complex 3D structures. This structural flexibility allows millions of organic molecules to exist.
However, not all carbon compounds are organic.
Carbonates, carbides, and carbon dioxide are classified as inorganic.
This classification is based on chemical structure and bonding patterns, not on whether a substance is natural or synthetic.
Understanding this distinction prevents confusion and improves conceptual clarity.
Organic compounds form the basis of organic chemistry, a major branch of chemistry focused on carbon-containing molecules and their reactions. Understanding organic chemistry helps explain biological processes, fuel combustion, and polymer production.
Why Carbon is Unique in Chemistry
Carbon’s importance comes from three key properties:
Tetravalency
Carbon forms four covalent bonds. This allows stable and diverse molecular structures.
Catenation
Carbon bonds with itself repeatedly, forming long chains and rings.
Multiple Bond Formation
Carbon forms single, double, and triple bonds.
No other element combines these features as effectively.
This is why organic chemistry is so vast.
This unique bonding ability explains why organic chemistry forms the foundation of biochemistry, polymer science, fuel chemistry, and many applied sciences.
Why Organic Compounds Matter
Every breath you take and every movement you make depends on organic molecules working inside your cells.
Organic compounds:
- Build cell membranes
- Store and release energy
- Carry genetic information
- Enable metabolism
- Form medicines and fuels
- Create polymers and synthetic materials
They connect biology with industry. Their real-world applications are visible in everyday products and processes, as discussed in the uses of organic compounds in daily life.
Nearly all biochemical reactions inside cells involve organic molecules interacting with each other.
Importance of Organic Compounds in Living Organisms
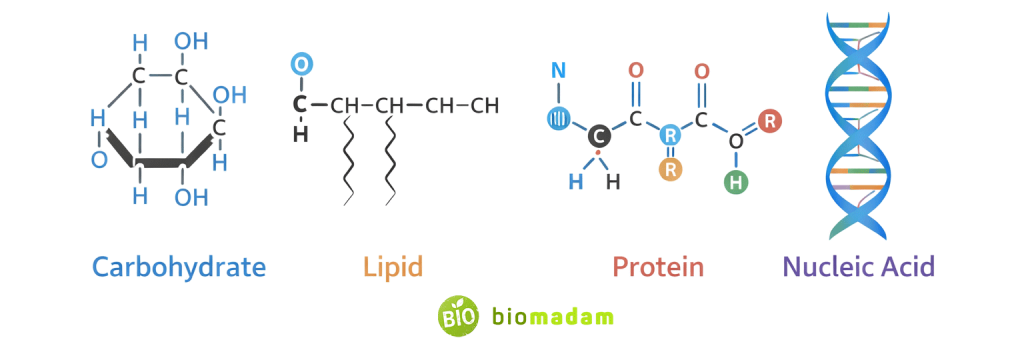
Living systems rely on four major classes of organic molecules.
Each class performs a distinct but interconnected function in maintaining life.
Carbohydrates – Immediate Energy Source
Carbohydrates contain carbon, hydrogen, and oxygen in a 1:2:1 ratio.
They:
- Provide rapid energy
- Maintain blood glucose levels
- Support structural integrity (cellulose in plants)
Glucose is the primary fuel of cells.
It provides about 4 kcal per gram.
Excess glucose is stored as glycogen in the liver and muscles.
When energy is needed, glycogen breaks down back into glucose.
This process maintains metabolic balance.
Lipids – Long-Term Energy & Structure
Lipids include fats, oils, waxes, and steroids.
They:
- Store concentrated energy
- Form cell membranes (phospholipids)
- Protect organs
- Act as hormones
Lipids provide approximately 9 kcal per gram.
They store more energy because they contain a higher number of carbon-hydrogen bonds. These bonds release large amounts of energy during oxidation.
This is why fat is the body’s long-term energy reserve.
Proteins – Structure, Function, and Enzymes
Proteins are made of amino acids.
They:
- Form muscles, skin, cartilage, and connective tissues
- Act as enzymes to speed up reactions
- Regulate hormones
- Support immune defense
Without enzymes, cellular reactions would occur too slowly to sustain life.
Proteins are not the primary energy source, but they can provide about 4 kcal per gram when needed.
Nucleic Acids – Genetic Control
DNA and RNA store and transmit genetic information.
They:
- Direct protein synthesis
- Control cell function
- Enable reproduction and inheritance
Without nucleic acids, biological continuity would be impossible.
Understanding these organic biomolecules is also critical in healthcare fields, including why chemistry is important in nursing.
Industrial and Agricultural Importance
Organic chemistry extends far beyond biology.
In agriculture:
- Organic fertilizers (manure, compost, urea) replenish soil nutrients.
- Organic-based pesticides and herbicides protect crops
- Soil organic matter improves structure and water retention
In industry:
- Petrochemicals are the backbone of plastic production
- Organic solvents are used in manufacturing
- Synthetic rubber, dyes, adhesives, and coatings depend on organic compounds, forming the material foundation discussed in the importance of chemistry in engineering.
- Pharmaceutical drugs are largely organic molecules
Modern industry runs on carbon chemistry.
Importance in the Fuel Industry
Many fuels are organic compounds.
Examples include:
- Methane (natural gas)
- Butane (LPG)
- Petrol and diesel (hydrocarbons)
These hydrocarbons release energy when burned.
That energy powers transportation, electricity generation, and manufacturing.
Organic chemistry directly fuels global economies.
Environmental and Sustainability Relevance
Organic compounds also play a major role in environmental science.
Biofuels such as ethanol and biodiesel are renewable organic fuels.
Biodegradable plastics are designed using specific organic polymers that break down more easily.
However, excessive combustion of fossil-fuel-based organic compounds contributes to greenhouse gas emissions.
Understanding organic chemistry helps develop greener technologies and sustainable materials.
The environmental impact depends on how organic compounds are produced, used, and managed.
The balance between synthetic organic production and sustainable green chemistry practices is now a major global research focus.
Comparison: Where Organic Compounds Matter Most
| Area | Role of Organic Compounds | Example Organic Compounds |
| Living Systems | Energy, structure, genetic control | Glucose, DNA, Proteins |
| Agriculture | Soil nutrients, pest control | Urea, Organic pesticides |
| Medicine | Drug design and therapy | Antibiotics, Hormones |
| Fuel Industry | Energy production | Methane, Petrol |
| Manufacturing | Plastics, polymers, solvents | Polyethylene, PVC |
What Would Happen Without Organic Compounds?
Without organic compounds:
- Cells could not form
- DNA and proteins would not exist
- Energy storage would be impossible
- Life as we know it would disappear
Modern medicine, fuels, plastics, and agriculture would collapse.
Organic compounds are not optional. They are foundational.
Common Misconceptions
Misconception 1: Organic means natural or chemical-free.
In chemistry, organic simply means carbon-based.
Misconception 2: All carbon compounds are organic.
Carbonates and carbon dioxide are inorganic.
Misconception 3: Organic compounds only exist in living organisms.
Many are synthetic and manufactured industrially.
Safety and Practical Implication
While organic compounds are essential, some can be hazardous.
Examples:
- Certain hydrocarbons are flammable
- Some synthetic organic chemicals may be toxic
- Improper pesticide use can harm ecosystems
Understanding organic chemistry improves safe handling and responsible use.
One-Line Summary
Organic compounds are carbon-based molecules essential for life, energy storage, medicine, agriculture, fuels, and modern industry.
FAQs
1. Why are organic compounds essential for life?
They form carbohydrates, proteins, lipids, and nucleic acids that sustain cellular structure and metabolism.
2. Why is carbon central to organic compounds?
Carbon forms four stable bonds and creates diverse molecular structures.
3. Are fuels organic compounds?
Most common fuels, including petrol and methane, are organic hydrocarbons.
4. Do organic compounds only exist naturally?
No. Many are synthetic and produced industrially.
5. Why are lipids more energy-dense than carbohydrates?
Lipids contain more carbon-hydrogen bonds, which release more energy during oxidation.
Why is organic chemistry important in industry?
Organic chemistry allows the design and production of fuels, plastics, pharmaceuticals, polymers, dyes, and synthetic materials used globally.
The Bottom Line
Organic compounds are the chemical backbone of life and the driving force of modern civilization. From your DNA to industrial fuels and pharmaceuticals, carbon-based molecules shape biological systems and global industries alike.

The Science Editorial Team creates clear, accurate, and student-friendly explanations of scientific concepts across biology, chemistry, physics, taxonomy, anatomy, and related life sciences. Content is written for educational and informational purposes using standard academic references and reliable sources. Read more about the → Science Editorial Team
Hi, I learned a lot… I need to know when this was published for my reference in my research.
Hey, hope you’re doing great!
The article was published on 6th Dec’22.