Every living body consists of numerous essential participants that play a pivotal role in bringing it up. Such a participant is an enzyme. Now, what are enzymes?
This group of biomolecules synthesizes in the living cells and act as catalysts throughout their entire life. All enzymes tend to accelerate a chemical reaction. We further classify them into six primary groups that will be discussed later on in this article. Before that, let’s talk about the basics of enzymes.

What Does An Enzyme Do?
The living bodies perform almost all of their functions with the help of enzymes. The renowned of all duties are:
- It helps in increasing the rate of reaction
- Moreover, enzymes do not participate in the chemical reaction but only accelerate them
Where Are Enzymes Made?
Scientists called enzymes biocatalysts as nature manufactures them in living cells. Every existing cell has ribosomes in it, either scattered or fixed to the rough endoplasmic reticulum. The primary function of these ribosomes is to prepare proteins, which are then called enzymes. It means all enzymes are proteins, but not all proteins are enzymes. Soon after they mature, these enzymes undergo post-translation modification, proceeding to another organelle called Golgi bodies.
Characteristics of Enzymes
Protein in Nature
Enzymes are the natural proteins found everywhere in a body and are primarily composed of globular proteins. Furthermore, these molecules vary with their entire protein weight so, specific for certain reactions.
An Enzyme Acts as a Catalyst
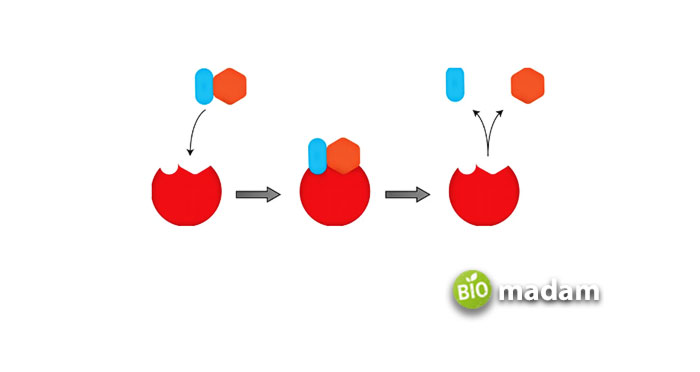
Enzymes generally act on a substance to convert it to a product. And almost all the living organisms’ phenomenon occurs with enzymes acting as a catalyst and converting the substrates to the relevant products.
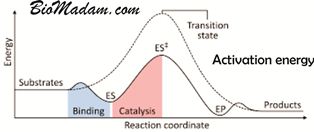
An Enzyme Accelerates the Reaction
Moreover, these enzymes speed up the reaction in the biological processes by reducing activation energy. It is the minimum energy required to kick start a chemical reaction. This activation energy can also help a single process run multiple times and regulate it.
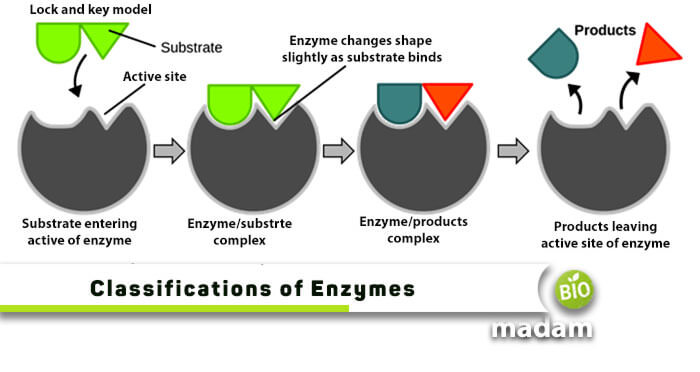
Lock And Key Model
Emil Fischer, in 1894, proposed a “lock and key model.” According to this model, an enzyme is a rigid structure, only precise to substrates matching its active site. It is similar to lock and key, as a particular key can only open a specific lock. In a word, enzymes are specific in nature.
Specific in Functioning
Most people widely use the term “One Enzyme One Substrate.” It means an enzyme works specifically for a substrate and is not available for many. So, at a time, a single enzyme catalyzes a single reaction.
Reverse Function
Enzymes work only to accelerate a reaction but never determine its directions or pathways. An enzyme will continue performing its function until the reaction reaches an equilibrium state. Thus, these molecules are highly explicit.
Minute Amount of Enzymes Required
Its fundamental concern is to speed up a reaction. Therefore a chemical reaction requires a minute amount of enzymes only.
How Do You Name Enzymes?
The general method of naming an enzyme first comprehends the substrate it is about to act on. Then, after observing the name of the substrate, a suffix “ase” adds at its end. For example, a catalyst oxidoreductase speeds up the oxidation-reduction reaction. It is one of the primary catalysts, falling in enzyme classification. Similarly, an enzyme lipase is responsible for hydrolyzing lipid molecules.
Classification of Enzymes
International Union of Biochemists classified enzymes into six primary types, depending upon where they act to catalyze a chemical reaction. Let’s briefly discuss them one by one:
Oxidoreductase
The enzyme which is involved in the oxidation and reduction reaction of its substrate is called oxidoreductase. For instance, xanthine oxidase, lactate dehydrogenase gG-6-P dehydrogenase, etc.
Transferase
Transferase molecules activate when a particular functional group transports from a donor to an acceptor molecule. For example, phosphoglucomutase, hexokinase, etc.
Hydrolases
We call them hydrolases as these associated with breaking a bond by adding water (through hydrolysis). For example, pepsin glucose-6- phosphatase and glycoside hydrolases.
Lyase
These are the group of enzymes that catalyzes a chemical reaction by forming double bonds. Lyases never tend to utilize oxidation or hydrolysis to accelerate a biochemical process. For instance, histidine decarboxylase, fumarase, etc.
Isomerase
This enzyme is responsible for isomerizing a substrate, thus altering its structure. Some examples include UDP- glucose, epimerase, retinal isomerase, etc.
Ligase
Another class of enzymes is Ligases. These involve the joining of two substrates with each other, such as glutamine synthase, RNA synthase. Besides, DNA ligases are also a prominent example, connecting DNA’s two fragments.
Proposed Models of Enzymes
Last but not least, there were two fundamental models of enzymes, so let’s briefly discuss them side by side:
Lock and Key Model
Emil Fischer first proposed this model in the late 1890s. He presented that the active site of an enzyme stands in proper confirmation. In other words, the active site of a catalyst provides a fixed and rigid shape where a substrate sets. The German chemist called it the lock and key model as only a definite key can open a specific lock. This model confirms that enzymes are distinct which can only act on a single substrate.
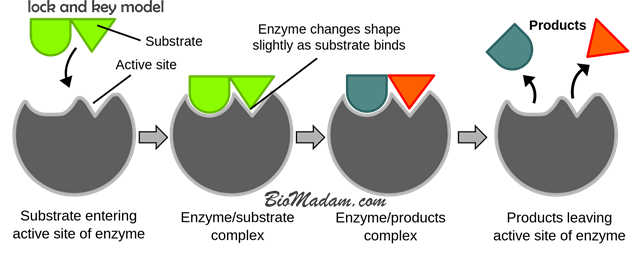
Induced Fit Model
Koshland, in 1958, presented Induced Fit Theory. This model completely contradicts the lock and key model, declaring that an enzyme is not a rigid structure and can be modified according to situations. In a particular chemical reaction involving enzymes, its active site can endure conformational alterations for augmented binding.
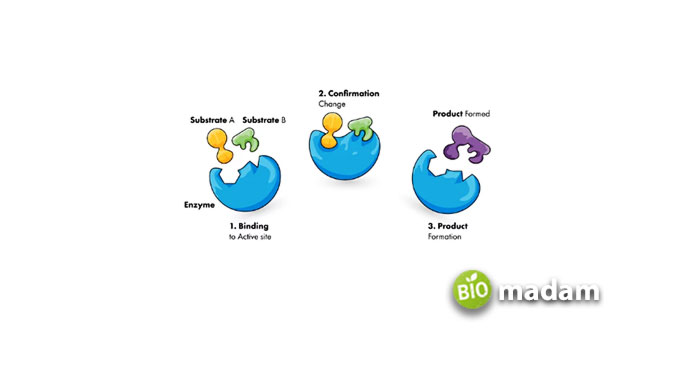
Nowadays, biologists prefer this model over the lock and key model due to its following advantages:
- The Induced Fit model offers a broad-spectrum enzyme specificity, e.g., a lipase enzyme can accelerate various lipid molecule reactions.
- The conformational variations can increase enzyme activity.
Conclusion
Enzymes are essential for many chemical reactions that keep living things alive. As explained in this article, they are very specific, efficient, and adaptable in speeding up these reactions. While scientists are still learning exactly how enzymes work, the two main models – lock-and-key and induced fit – help us understand their structure and what they do. More research on enzymatic structure, control, and speed of reactions will increase our knowledge about these biomolecules. For now, we can appreciate that even the most basic life functions depend on the hard work and functioning of enzymes.
FAQs
What is a co-enzyme?
Sometimes an enzyme needs an external aid to work efficiently, for which it uses a co-enzyme or a co-substrate. This structure makes a bond with the enzyme molecule and leaves it at the end of the chemical reaction soon after the process completes. A coenzyme is named so because it makes an early bond with the enzyme than the substrate itself. It is usually an organic, non-protein molecule that binds to the protein part of an enzyme. Furthermore, these activated vitamins make complexes for running metabolic pathways and transform nutrients into vital energy.
Differentiate between an apoenzyme and a holoenzyme?
Apoenzymes are the intact protein part of an enzyme, lying in their inactive form. On the contrary, a holoenzyme is a complex constituted by an apoenzyme and a co-factor. An apoenzyme triggers only when attached to a co-factor (non-protein factor). In contrast, a holoenzyme always stays in its active form to accelerate a biochemical reaction. Examples of apoenzymes include carbonic anhydrase, pepsin, etc. Similarly, some of the holoenzymes are DNA & RNA polymerases.
What is a zymogen?
We can also pronounce zymogens as proenzymes. This group of enzymes usually does not remain in their active form but can be converted if needed. A zymogen activates when another enzyme from within the organism triggers it up. Zymogens function to stop excessive protein degradation.

Hello, I would like to introduce myself to you! I am Chelsea Rogers, an experienced blog writer for science articles, holding an MPhil degree. My enthusiasm to grab the best knowledge, let it relate to botany, zoology, or any other science branch. Read my articles & let me wait for your words s in the comment section.