Quick Answer: The main difference between alpha and beta decay is the type of particle emitted from an unstable nucleus.
Alpha decay releases an alpha particle (two protons and two neutrons), decreasing the atomic number by 2 and mass number by 4.
Beta decay releases a beta particle (electron or positron), changing the atomic number by ±1 while the mass number remains unchanged.
Comparison Between Alpha and Beta Decay
| Feature | Alpha Decay | Beta Decay |
| Emitted Particle | Alpha particle (⁴₂He nucleus) | Beta particle (electron β⁻ or positron β⁺) |
| Change in Atomic Number | −2 | +1 (β⁻) or −1 (β⁺) |
| Change in Mass Number | −4 | No change |
| Occurs In | Very heavy nuclei (e.g., uranium) | Neutron-rich or proton-rich nuclei |
| Penetration Power | Low (stopped by paper) | Moderate (stopped by thin aluminum) |
| Ionizing Power | High | Moderate |
| Speed | ~5–10% speed of light | Close to speed of light |
| Example | ²³⁸U → ²³⁴Th + ⁴He | ²³⁴Th → ²³⁴Pa + β⁻ |
What is Radioactive Decay?
Radioactive decay is the spontaneous transformation of an unstable atomic nucleus into a more stable nucleus by emitting radiation. This process was discovered by Henri Becquerel in 1896, marking the beginning of nuclear physics research.
Radioactive decay is governed by fundamental nuclear forces and follows conservation laws of energy and momentum. It is a central topic in nuclear physics, a major branch of Physics that examines the structure and behavior of atomic nuclei.
There are three primary types of radioactive decay:
- Alpha decay
- Beta decay
- Gamma decay
This article focuses specifically on alpha and beta decay.
What is Alpha Decay?
Alpha decay occurs when an unstable heavy nucleus emits an alpha particle, which consists of two protons and two neutrons (equivalent to a helium-4 nucleus).
Nuclear Equation Example
²³⁸₉₂U → ²³⁴₉₀Th + ⁴₂He
Another Example of Alpha Decay
²²⁶₈₈Ra → ²²²₈₆Rn + ⁴₂He
Radium transforms into radon by emitting an alpha particle.
What Happens During Alpha Decay?
- Atomic number decreases by 2
- Mass number decreases by 4
- A new element forms (transmutation occurs)
Why Does Alpha Decay Occur?
It typically occurs in very heavy nuclei where strong electrostatic repulsion between protons makes the nucleus unstable. Emitting an alpha particle reduces this repulsion.
Properties of Alpha Particles
- Heavy and positively charged
- Low penetration power (blocked by paper or skin)
- High ionizing ability
- Travel a few centimeters in air
What is Beta Decay?
Beta decay occurs when a neutron or proton inside the nucleus transforms to achieve stability.
There are two types:
1. Beta Minus (β⁻) Decay
Occurs in neutron-rich nuclei.
Process:
A neutron converts into:
- A proton
- An electron (β⁻)
- An antineutrino
Example:
²³⁴₉₀Th → ²³⁴₉₁Pa + β⁻ + ν̅
Result:
- Atomic number increases by 1
- Mass number remains unchanged
2. Beta Plus (β⁺) Decay
Occurs in proton-rich nuclei.
Process:
A proton converts into:
- A neutron
- A positron (β⁺)
- A neutrino
Result:
- Atomic number decreases by 1
- Mass number remains unchanged
Example of Beta Plus (β⁺) Decay
²²₁₁Na → ²²₁₀Ne + β⁺ + ν
Sodium-22 undergoes positron emission, decreasing its atomic number by one.
Key Concept: Why Alpha and Beta Decay Occur
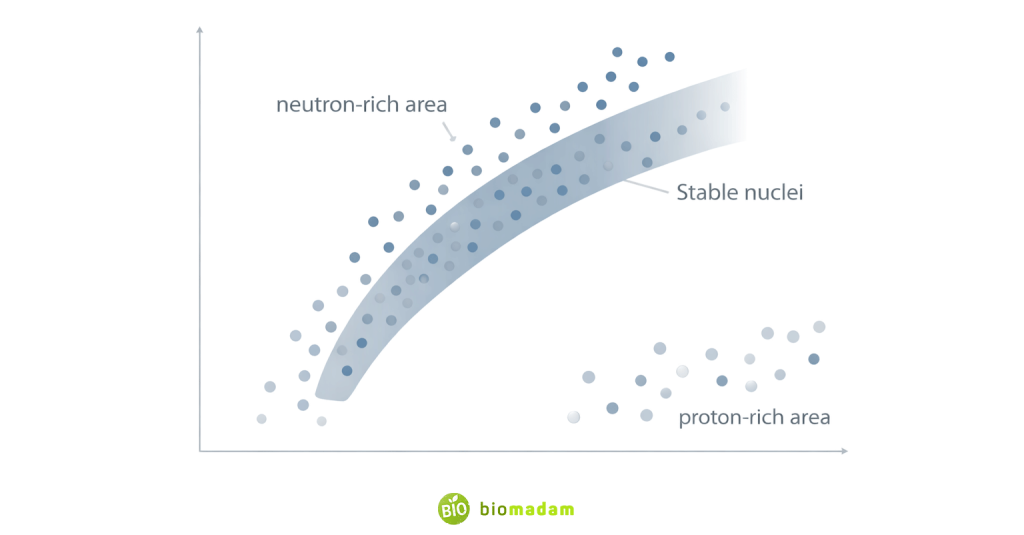
Atomic nuclei remain stable only within a certain neutron-to-proton (N/Z) ratio, often called the band (or valley) of stability. Nuclei outside this stability region undergo radioactive decay to restore balance.
Alpha Decay and Heavy Nuclei
Very heavy elements (typically atomic number > 83) contain:
- Large numbers of protons
- Strong electrostatic repulsion between protons
To reduce this repulsive force and move toward stability, the nucleus ejects a tightly bound alpha particle (⁴₂He nucleus). This significantly lowers both mass and atomic number.
Beta Decay and Neutron–Proton Imbalance
Beta decay corrects imbalance in the neutron-to-proton ratio:
- Neutron-rich nuclei → β⁻ decay (neutron converts to proton)
- Proton-rich nuclei → β⁺ decay (proton converts to neutron)
This adjustment moves the nucleus closer to the band of stability.
Gamma Decay Overview
Gamma decay occurs when an excited nucleus releases excess energy in the form of electromagnetic radiation without changing its atomic or mass number.
To understand how gamma radiation differs from particle emissions like alpha and beta radiation, see Difference Between X-Rays and Gamma Rays, which explains radiation origin and energy differences in detail.
Energy and Penetration Comparison
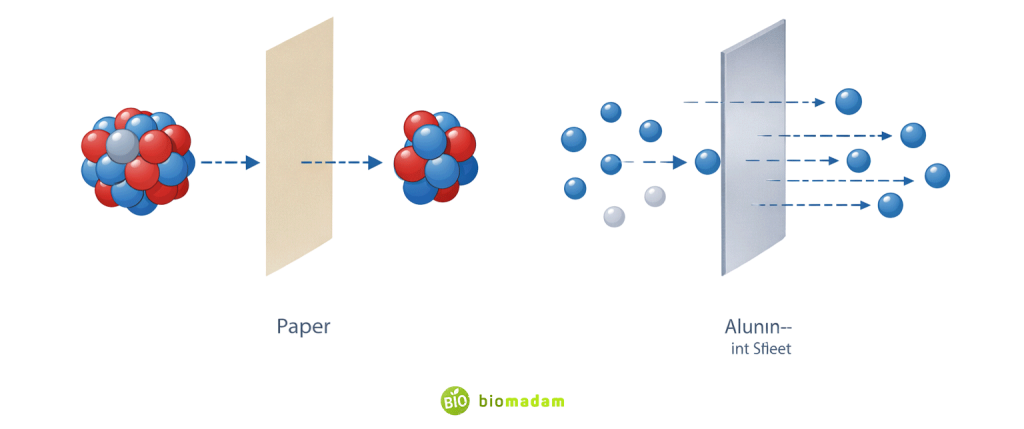
| Property | Alpha | Beta |
| Ionization | Very High | Moderate |
| Penetration | Very Low | Moderate |
| Shielding Material | Paper, skin | Aluminum sheet |
| Hazard Type | Dangerous if ingested | Can penetrate skin |
Energy Release and Q-Value in Alpha and Beta Decay
The energy released during radioactive decay is called the Q-value.
Alpha Decay Energy
Alpha decay releases a discrete (fixed) amount of energy, meaning emitted alpha particles have specific kinetic energies. This happens because it is a two-body decay process.
Beta Decay Energy
Beta decay produces a continuous energy spectrum. This occurs because the decay involves three particles:
- Beta particle
- Neutrino (or antineutrino)
- Daughter nucleus
The energy is shared among them, resulting in variable beta particle energies.
This difference is an important concept in nuclear physics.
Similarities Between Alpha and Beta Decay
- Both are forms of radioactive decay
- Both occur in unstable nuclei
- Both result in transmutation of elements
- Both release energy
Real-World Applications of Alpha and Beta Decay
Applications of Alpha Decay
- Smoke detectors use Americium-241, an alpha emitter.
- Radioisotope thermoelectric generators (RTGs) use alpha decay to produce power for spacecraft.
- Used in targeted cancer therapy research.
Applications of Beta Decay
- Carbon-14 dating relies on beta decay to determine the age of fossils.
- Medical tracers in diagnostic imaging.
- Radiation therapy for treating certain cancers.
Understanding these applications shows why radioactive decay is important in science, medicine, and technology.
Common Misconceptions About Alpha and Beta Decay
- Beta decay does NOT involve orbital electrons; it occurs inside the nucleus.
- Alpha particles are helium nuclei, not neutral helium atoms (they gain electrons later).
- Beta decay does NOT change the mass number.
- Alpha radiation is not highly penetrating but can be dangerous if inhaled or ingested.
Clarifying these misconceptions improves understanding of nuclear processes.
FAQs
Which is more dangerous, alpha or beta radiation?
Alpha radiation is more dangerous inside the body due to high ionization, while beta radiation is more penetrating externally.
Does beta decay change atomic mass?
No. The mass number remains the same in beta decay.
Why does alpha decay occur in heavy elements?
Heavy elements have strong proton repulsion, and emitting an alpha particle reduces instability.
Can alpha particles penetrate human skin?
No, alpha particles cannot penetrate skin but are harmful if inhaled or ingested.
Is beta decay faster than alpha decay?
Beta particles travel faster, but decay rate depends on the specific isotope.
Scientific Comparison Summary (For Quick Revision)
- Alpha decay → Heavy nuclei → Mass −4, Atomic number −2
- Beta minus decay → Neutron-rich nuclei → Atomic number +1
- Beta plus decay → Proton-rich nuclei → Atomic number −1
- Alpha particles → High ionization, low penetration
- Beta particles → Moderate ionization, moderate penetration
Conclusion
Alpha and beta decay are fundamental nuclear processes that stabilize unstable nuclei through particle emission. Alpha decay reduces both mass and atomic number in heavy elements, while beta decay adjusts the neutron-to-proton ratio without changing mass number. Together, these mechanisms help unstable nuclei move toward stability while conserving fundamental physical quantities. Understanding alpha and beta decay is essential for nuclear physics, medical science, and radiation safety.

The Science Editorial Team creates clear, accurate, and student-friendly explanations of scientific concepts across biology, chemistry, physics, taxonomy, anatomy, and related life sciences. Content is written for educational and informational purposes using standard academic references and reliable sources. Read more about the → Science Editorial Team
