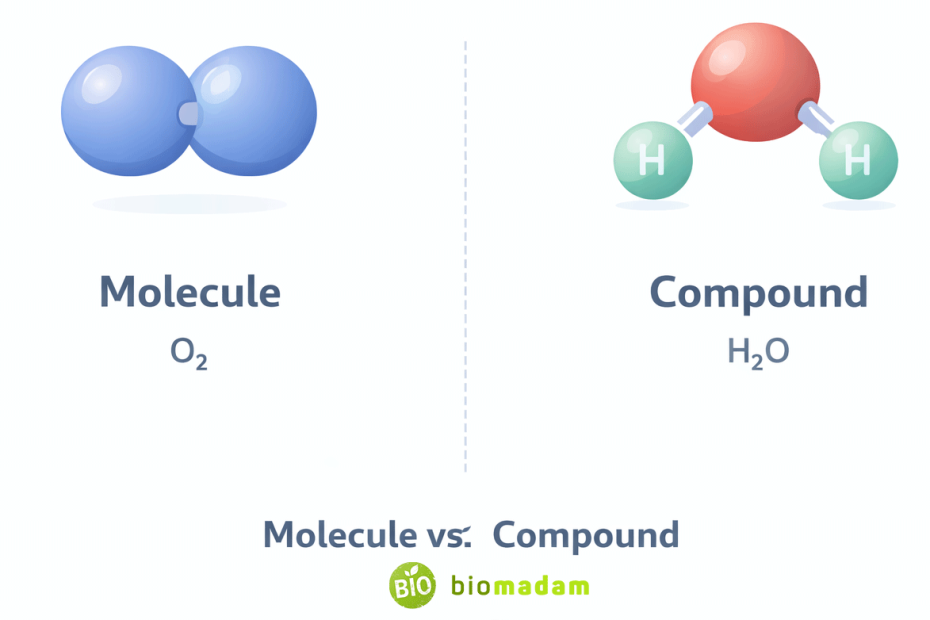
Quick Explanation: A molecule is a group of two or more atoms chemically bonded together that acts as a single unit. A compound is a type of molecule that contains atoms of two or more different elements chemically bonded in a fixed ratio.
The key distinction: Most covalent compounds exist as discrete molecules, but not all molecules are compounds..
Core Concept First: What Actually Separates Them?
The real difference lies in elemental composition:
- A molecule can consist of the same element (e.g., O₂).
- A compound must contain different elements (e.g., H₂O).
So, the deciding factor is not bonding type, stability, or visibility — it is whether different elements are present.
Comparison Between Molecules and Compounds
| Feature | Molecule | Compound |
| Definition | Two or more atoms chemically bonded | Two or more different elements chemically bonded |
| Element Type | Same or different elements | Always different elements |
| Smallest Unit | Smallest independent unit of a substance | Smallest representative unit of a pure compound |
| Composition | May be homonuclear or heteronuclear | Always heteronuclear |
| Example | O₂, N₂, O₃ | H₂O, CO₂, NaCl (formula unit) |
| Relationship | Broader term | Specific type of molecule |
Molecule vs Compound in One Line
- Molecule → atoms bonded together
- Compound → atoms of different elements bonded together
What is a Molecule?
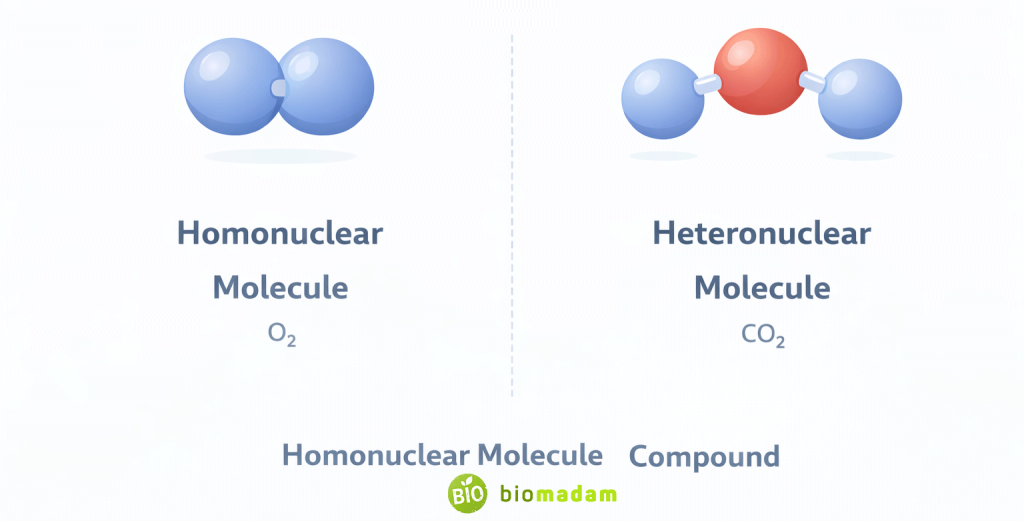
A molecule is formed when atoms join together through chemical bonding (typically covalent bonding) to form the smallest unit that retains the substance’s chemical properties. The distinctions among bond types are explained in the Difference between Ionic, Covalent and Metallic Bonds.
Key points:
- Can contain identical atoms (O₂, N₂)
- Can contain different atoms (H₂O, NH₃)
- Represented by chemical formulas
- May be neutral or charged (molecular ions exist)
The nature of charged species is discussed further in the Difference Between Cations and Anions.
Examples:
- Oxygen gas (O₂)
- Ozone (O₃)
- Water (H₂O)
Molecules can exist independently in nature.
A compound is a pure substance formed when atoms of two or more different elements chemically combine in a fixed ratio. In classification of matter, compounds are categorized as pure substances, as explained further in the Difference Between Pure Substance and Mixture.
Compounds differ from physical combinations of substances because chemical bonding creates a new chemical identity. This distinction is discussed in more detail in the Difference Between Compounds and Mixtures.
Key points:
- Always contains different elements
- Has fixed composition by mass
- Compounds follow the Law of Definite Proportions, meaning their elements combine in fixed mass ratios regardless of sample size
- Has distinct physical and chemical properties
Examples:
- Water (H₂O)
- Carbon dioxide (CO₂)
- Sodium chloride (NaCl)
Note: Some ionic compounds (like NaCl) form lattice structures rather than discrete molecules. In such cases, the smallest unit is called a formula unit, not a molecule.
Important Clarification: Molecule vs Ionic Compound
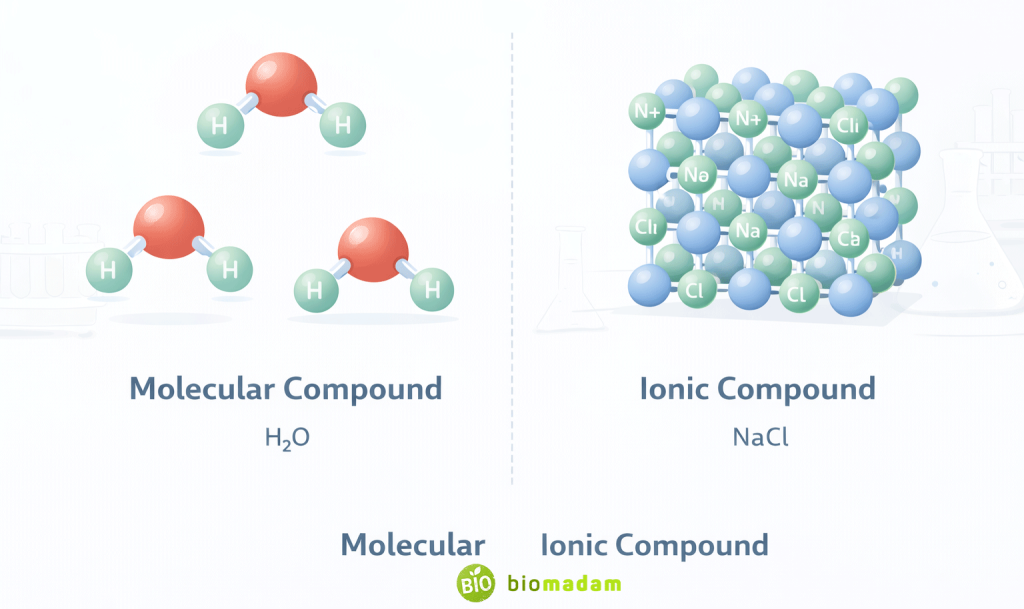
Not all compounds exist as discrete molecules.
For example:
- Water (H₂O) → molecular compound
- Sodium chloride (NaCl) → ionic compound (crystal lattice)
NaCl is a compound but not a molecule in solid form.
This distinction prevents a common misconception.
Structural Perspective
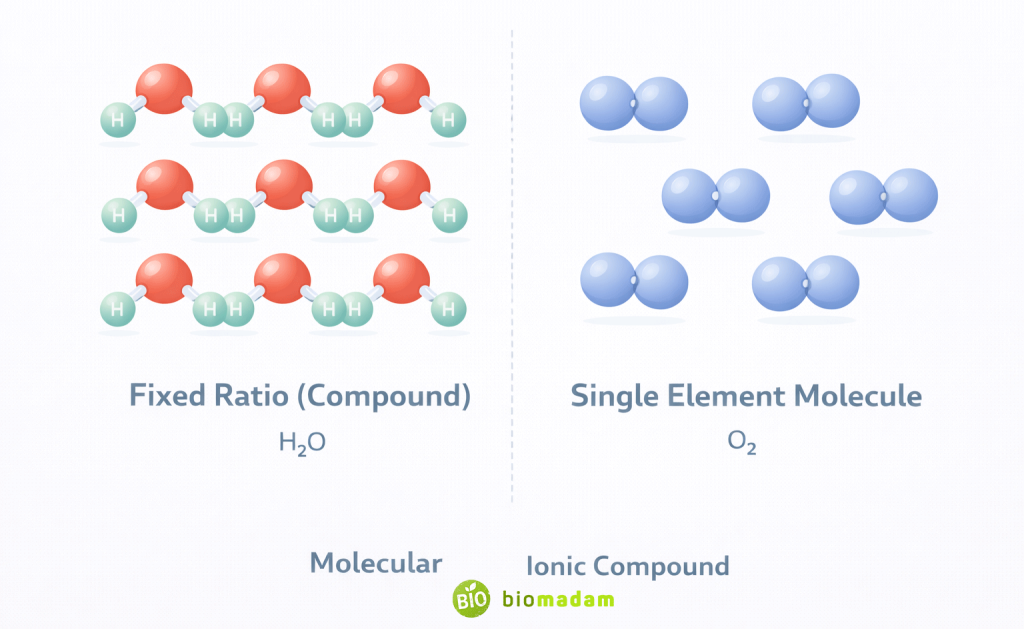
Think of it this way:
- Molecule → Focuses on bonding between atoms.
- Compound → Focuses on chemical composition involving different elements.
So molecule describes structure.
Compound describes composition.
Common Confusion Cases
Is O₂ a compound?
No. O₂ is a molecule because it contains only oxygen atoms.
Is H₂O a molecule?
Yes. It is both a molecule and a compound.
Is NaCl a molecule?
In solid form, no. It is an ionic compound made of repeating formula units.
Key Differences Summary
- Molecule → two or more atoms bonded
- Compound → two or more different elements bonded
- Most covalent compounds exist as molecules, while ionic compounds exist as repeating formula units.
- Not all molecules are compounds
- Ionic compounds may not form discrete molecules
Real-Life Examples Explained
- Oxygen gas (O₂) → molecule but not a compound
- Water (H₂O) → both a molecule and a compound
- Sodium chloride (NaCl) → compound but not a discrete molecule in solid form
FAQs
Can a molecule exist without being a compound?
Yes. Molecules like O₂ and N₂ consist of only one element, so they are not compounds.
Are all compounds molecules?
Most covalent compounds are molecules. However, ionic compounds form formula units rather than discrete molecules.
What is the simplest example of a molecule?
Oxygen gas (O₂) is a simple diatomic molecule.
What makes a compound different from an element?
A compound contains atoms of different elements chemically bonded, while an element contains only one type of atom.
Is ozone a compound?
No. Ozone (O₃) is a molecule made of only oxygen atoms, so it is not a compound.
Are molecular elements compounds?
No. Elements such as O₂, N₂, and S₈ form molecules but are not compounds because they contain only one type of atom.
The Bottom Line
A molecule refers to atoms chemically bonded together, regardless of whether they are the same or different elements. A compound is a specific type of molecule composed of different elements in fixed ratios. Understanding this distinction clarifies many foundational chemistry concepts related to bonding, classification of matter, and chemical identity.

The Science Editorial Team creates clear, accurate, and student-friendly explanations of scientific concepts across biology, chemistry, physics, taxonomy, anatomy, and related life sciences. Content is written for educational and informational purposes using standard academic references and reliable sources. Read more about the → Science Editorial Team