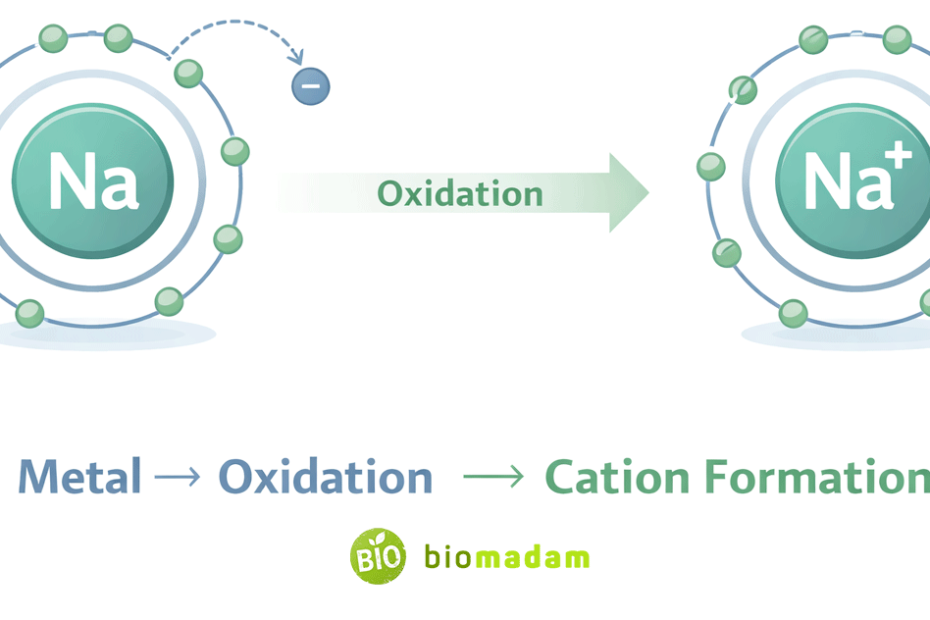
Quick Answer: Metals form cations, not anions.
In standard chemical reactions, metals form positive ions (cations), not negative ions (anions).
They lose one or more electrons from their outer shell and become positively charged ions.
In simple terms: Metal − electron(s) → Positive ion (cation)
Core Concept: Why Metals Lose Electrons
Atoms are neutral when:
- Number of protons = Number of electrons
Metals usually have 1–3 valence electrons.
These outer electrons are loosely held.
Because of this:
- Metals lose electrons easily
- When metals lose electrons, they undergo oxidation, which results in the formation of a cation.
- Losing electrons gives them a positive charge
- The result is a cation
Example:
Na → Na⁺ + e⁻
Sodium loses one electron and becomes positively charged.
What Are Metals?
Metals are elements found mostly on the left and center of the periodic table.
Common properties:
- Shiny (lustrous)
- Good conductors of heat and electricity
- Malleable and ductile
- Usually solid at room temperature (except mercury)
Common metals include:
- Sodium (Na)
- Calcium (Ca)
- Magnesium (Mg)
- Aluminum (Al)
- Iron (Fe)
- Potassium (K)
Most metals are found in nature as ores.
What Are Cations and Anions?
When atoms gain or lose electrons, they form ions, details are explained in Difference Between Cations and Anions
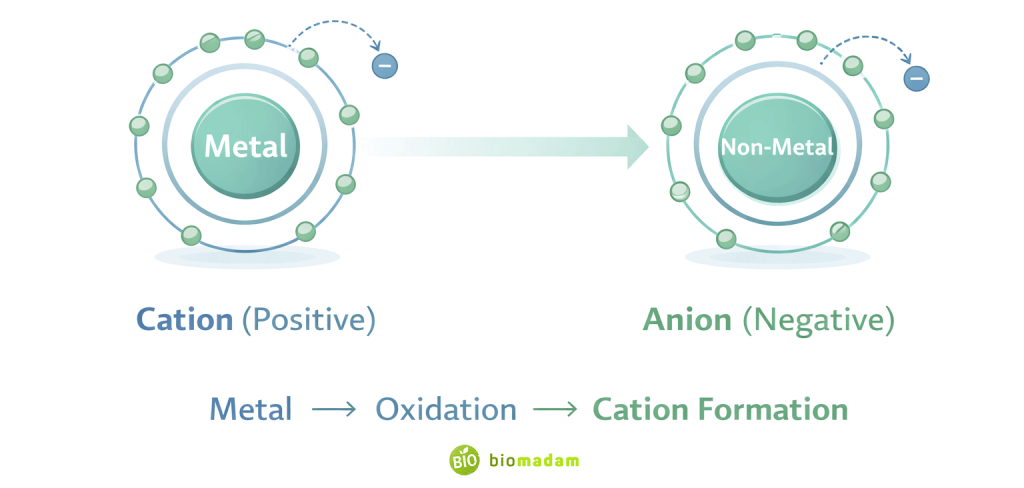
Cations
- Positively charged
- Formed by loss of electrons
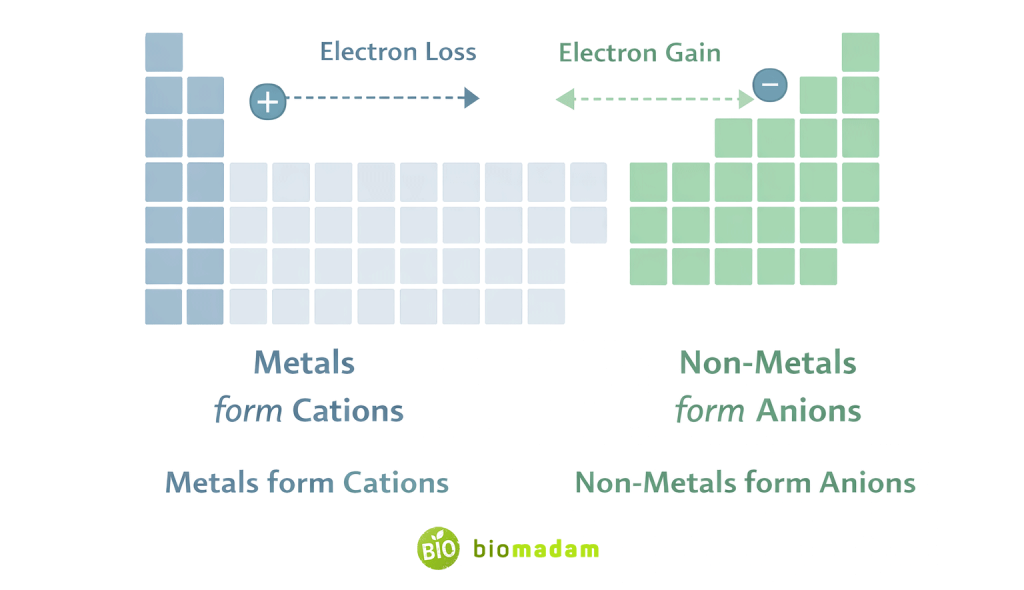
- Usually formed by metals
The detailed mechanism of positive ion formation is explained in How Are Cations Formed?
Anions
- Negatively charged
- Formed by gain of electrons
- Usually formed by non-metals
Example:
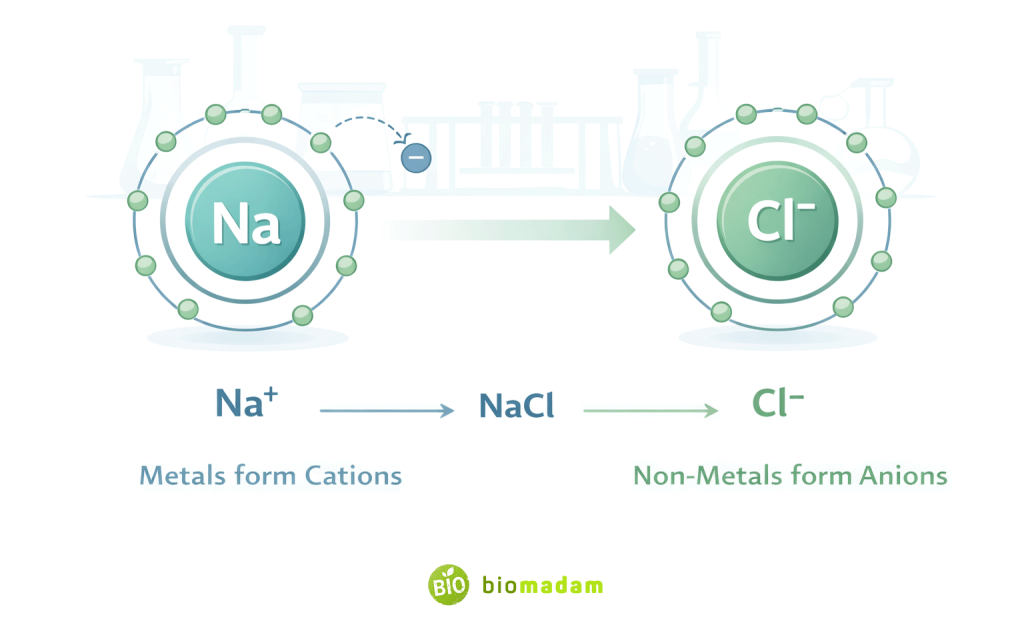
Na → Na⁺ (cation)
Cl + e⁻ → Cl⁻ (anion)
Do Metals Form Anions or Cations?
Metals form cations.
They do this because:
- They generally have relatively low ionization energy compared to non-metals.
- They hold outer electrons weakly
- They become more stable after losing electrons
Non-metals, on the other hand:
- Have high electron affinity
- Gain electrons
- Form anions
When a metal and non-metal react, they form an ionic compound, a bonding type explained in the Difference between Ionic, Covalent and Metallic Bonds.
Example:
Na⁺ + Cl⁻ → NaCl
The positive and negative ions attract each other.
In electrochemical cells and batteries, metals form cations at the anode during oxidation reaction, explained further in the Difference Between Anode and Cathode.
Cations vs Anions (Quick Comparison)
| Property | Cations | Anions |
| Charge | Positive | Negative |
| Formed By | Metals | Non-metals |
| Electron Change | Lose electrons | Gain electrons |
| Attracted To | Cathode | Anode |
| Size | Smaller than atom | Larger than atom |
| Example | Na⁺, Ca²⁺ | Cl⁻, Br⁻ |
Why Do Metals Prefer to Form Cations?
It comes down to electron configuration.
Atoms want a stable outer shell.
Metals:
- Have few valence electrons
- Achieve stability by losing them
- Often reach a noble gas configuration
Example:
- Na (2,8,1) → loses 1 electron → becomes stable (2,8)
This stability drives cation formation.
Periodic Trend Insight
Metallic character increases down a group and decreases across a period in the periodic table.
The stronger the metallic character, the more easily an element loses electrons and forms cations.
This explains why alkali metals are highly reactive and readily form +1 ions.
Can Metals Ever Form Anions?
In typical chemical reactions, metals form cations.
The formation of metal anions is extremely rare and not encountered in standard introductory chemistry.
In standard chemistry, metals form cations.
How to Predict If an Element Forms Cation or Anion
Look at its position in the periodic table.
- Group 1 (Alkali metals) → Always +1
- Group 2 (Alkaline earth metals) → Always +2
- Transition metals → Variable positive charges
- Halogens (Group 17) → Usually −1
- Noble gases (Group 18) → Rarely form ions
How to Write Ionic Formulas
Always write:
Cation first → Anion second
Example:
Na⁺ + Cl⁻ → NaCl
Charges are written as superscripts:
- Ca²⁺
- Mg²⁺
- Al³⁺
Common Misconception
Metals do not gain protons to become positive.
Ion formation involves electrons only.
Proton number changes only in nuclear reactions, not chemical reactions.
One-Line Summary
Metals form cations because they lose electrons easily and become positively charged to achieve stability.
FAQs
Do all metals form cations?
Yes. In normal chemical reactions, metals form positive ions.
Why don’t metals form anions?
Because they lose electrons easily instead of gaining them.
Can metals gain electrons?
In normal chemistry, metals rarely gain electrons because their low electronegativity favors electron loss over electron gain.
Why are metal ions smaller than their atoms?
When a metal loses electrons, it may lose part of its outer energy level. This reduces electron-electron repulsion and makes the resulting cation smaller than the original atom.
Which metals form more than one cation?
Many transition metals, such as iron (Fe²⁺ and Fe³⁺).
Do noble gases form ions?
No. Noble gases are stable and rarely form ions.
Metal Ion Formation in One Line
Metal → loses electron(s) → oxidation → positive charge → cation
The Bottom Line
Metals form cations by losing electrons.
This happens because their outer electrons are weakly held and can be removed easily.
Non-metals gain electrons and form anions.
Understanding this concept explains ionic bonding, compound formation, and periodic trends in chemistry.

The Science Editorial Team creates clear, accurate, and student-friendly explanations of scientific concepts across biology, chemistry, physics, taxonomy, anatomy, and related life sciences. Content is written for educational and informational purposes using standard academic references and reliable sources. Read more about the → Science Editorial Team