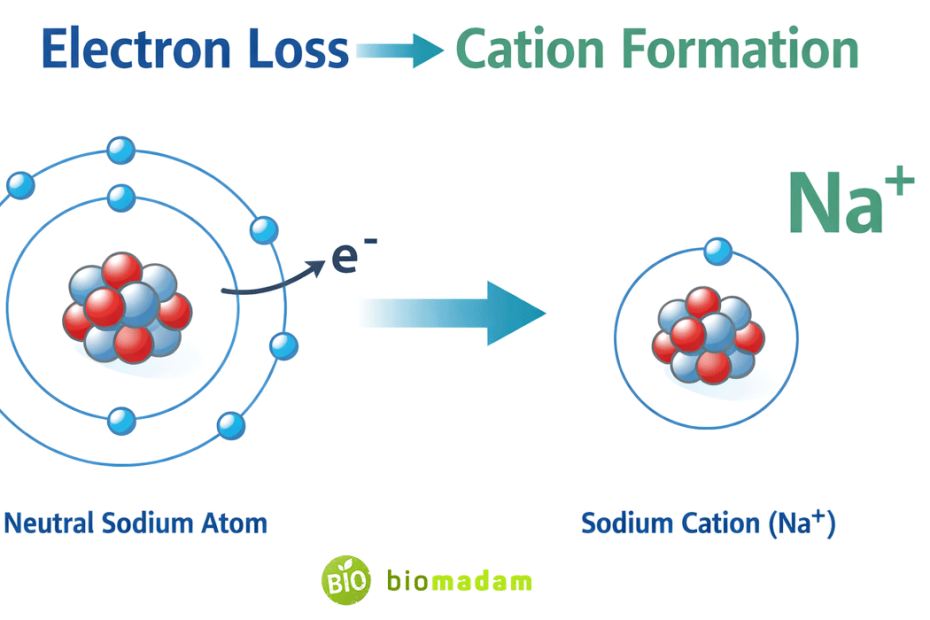
Quick Answer: Cations are formed when an atom or molecule loses one or more electrons during oxidation or ionization, resulting in a net positive charge because the number of protons exceeds the number of electrons.
Cation formation commonly occurs through oxidation, ionization, or electron transfer reactions.
Cation formation is a fundamental concept in chemistry when studying atomic structure and ionic bonding behavior.
Core Concept: Why Does Losing Electrons Create a Positive Charge?
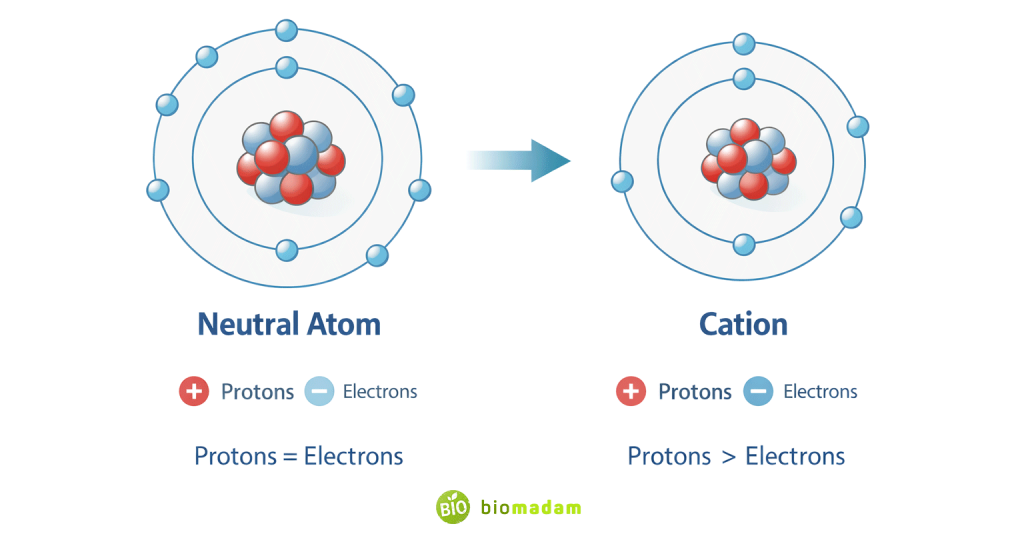
Atoms are electrically neutral when:
- Number of protons = number of electrons
When an atom loses an electron:
- Protons remain unchanged
- Electrons decrease
Since protons are positively charged and electrons are negatively charged, losing electrons causes:
Net Charge = Protons − Electrons → Positive value
Example:
- Na (11 protons, 11 electrons)
After losing one electron:
- Na⁺ (11 protons, 10 electrons)
Step-by-Step Formation of a Cation
- A neutral atom contains equal numbers of protons and electrons.
- Energy or a chemical reaction removes one or more electrons.
- The number of protons becomes greater than the number of electrons.
- The atom acquires a net positive charge and becomes a cation.
Example:
- Na → Na⁺ + e⁻
Cation Formation in One Line
Neutral atom − electron(s) → Positive ion (cation)
Example:
- Na → Na⁺ + e⁻
What is a Cation?
A cation is a positively charged ion formed when an atom or molecule loses electrons.
Cations can be:
- Monatomic (Na⁺, Ca²⁺)
- Polyatomic (NH₄⁺)
Cations are commonly formed by metals, especially those in:
- Group 1 (Alkali metals)
- Group 2 (Alkaline earth metals)
- Transition metals
Main Processes That Form Cations
Oxidation (Electron Loss)
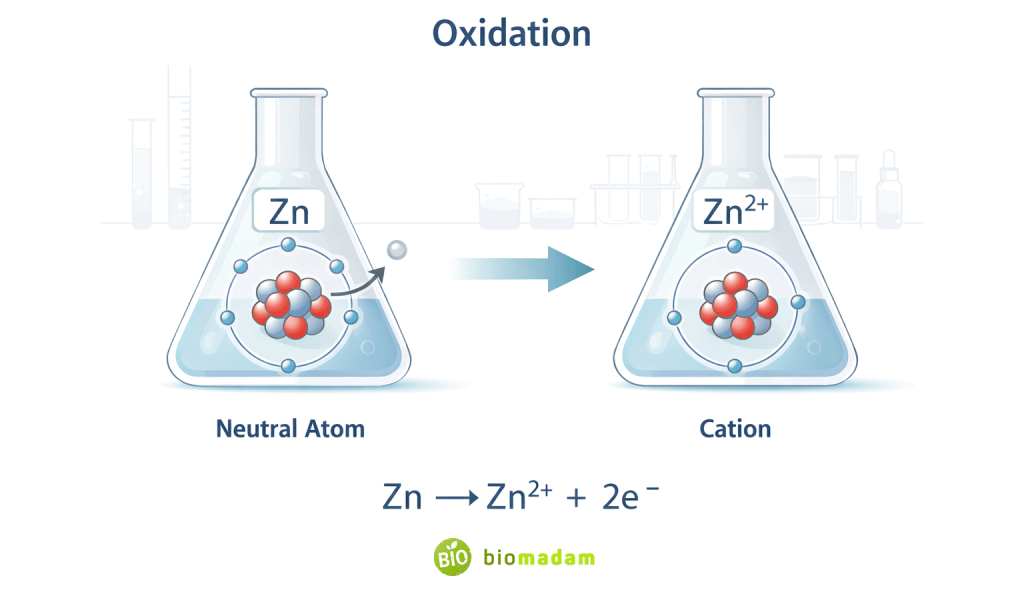
Oxidation is defined as loss of electrons.
When a metal undergoes oxidation, it forms a cation.
Example:
- Zn → Zn²⁺ + 2e⁻
In redox reactions:
- Oxidation → electron loss → cation formation
- Reduction → electron gain → anion formation
Electron Transfer Reactions
In ionic bond formation, electrons are transferred from one atom to another, as explained in Difference between Ionic, Covalent and Metallic Bonds.
Example:
- Na + Cl → Na⁺ + Cl⁻
Sodium loses one electron and becomes a cation.
Real-World Example
When sodium reacts with chlorine to form sodium chloride (NaCl), sodium loses one electron and becomes Na⁺, while chlorine gains an electron to become Cl⁻. The oppositely charged ions then attract to form an ionic compound.
Ionization (Energy-Induced Electron Removal)
Ionization occurs when energy (heat, radiation, or electrical energy) removes an electron from an atom.
In strict chemical terms, ionization energy refers to removing an electron from a gaseous atom.
Example:
- Na → Na⁺ + e⁻ (requires ionization energy)
The ionization energy is the energy required to remove an electron from a gaseous atom.
Ionization energy generally increases across a period and decreases down a group in the periodic table.
How Are Cations Formed from Elements?
Cations are formed from neutral elements when the element loses one or more valence electrons. This typically occurs with metals, which have low ionization energies and achieve stability by losing outer-shell electrons.
For example:
- Na (neutral element) → Na⁺ + e⁻
The element sodium loses one valence electron and becomes a cation.
How Are Cations Formed in Terms of Electrons?
In terms of electrons, cations form when the total number of electrons becomes less than the number of protons.
Since:
- Protons = positive charge
- Electrons = negative charge
Losing electrons results in a net positive charge.
Example:
- 11 protons − 10 electrons = +1 charge (Na⁺)
Why Metals Easily Form Cations
Metals tend to:
- Have 1–3 valence electrons
- Possess low ionization energy
- Achieve stability by losing electrons
When outer electrons are loosely held, they are easier to remove.
Example:
- Na forms Na⁺
- Ca forms Ca²⁺
- Al forms Al³⁺
For a broader explanation of metal ion behavior, see Do Metals Form Anions or Cations?
Cations in Electrochemistry
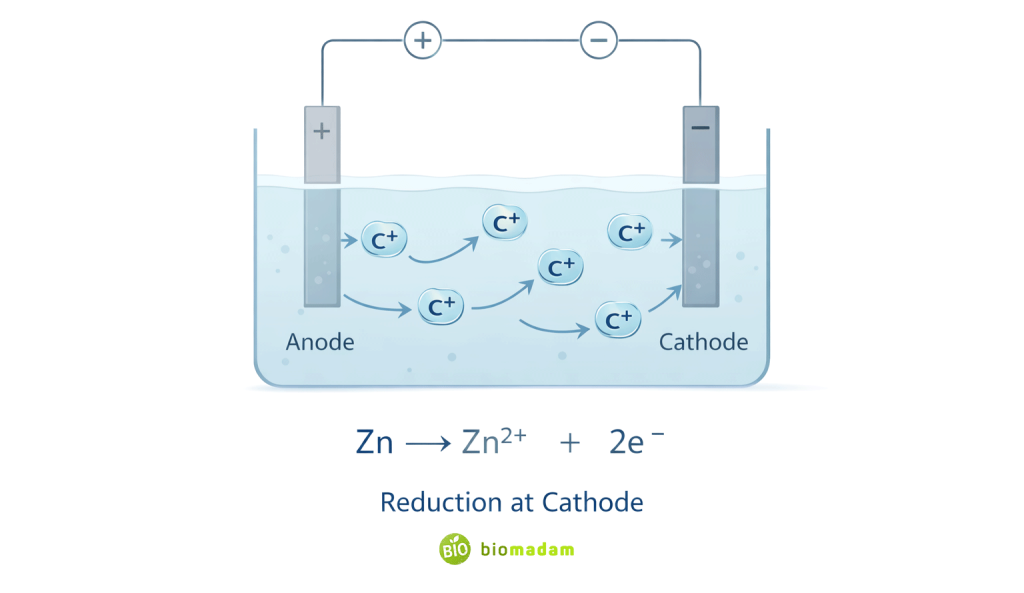
Cations move toward the cathode because reduction occurs there, and positively charged ions are attracted to the negatively charged region where they gain electrons.
In batteries:
- Oxidation occurs at the anode
- Cations migrate through the electrolyte toward the cathode
Common Examples of Cations
| Element | Cation Formed |
| Sodium | Na⁺ |
| Potassium | K⁺ |
| Calcium | Ca²⁺ |
| Magnesium | Mg²⁺ |
| Aluminum | Al³⁺ |
Some elements can form more than one cation.
Example:
- Fe → Fe²⁺ or Fe³⁺ depending on the reaction conditions.
This variability is common among transition metals.
Biological Importance of Cations
Cations are essential in biological systems:
- Na⁺ and K⁺ regulate nerve impulses
- Ca²⁺ controls muscle contraction
- Mg²⁺ stabilizes ATP in cells
- H⁺ plays a role in pH balance
The sodium-potassium pump maintains cellular ion balance.
Cation vs Anion
- Cation → loses electrons → positive charge
- Anion → gains electrons → negative charge
See Difference Between Cations and Anions
Common Misconception
Cations are not formed by gaining protons. In ordinary chemical reactions, ion formation involves the transfer or removal of electrons only. Changes in proton number occur only in nuclear reactions, not in chemical processes.
FAQs
Do all metals form cations?
Most metals form cations because they tend to lose electrons to achieve stable configurations.
Can nonmetals form cations?
Rarely under normal conditions. Nonmetals typically gain electrons and form anions.
What is ionization energy?
Ionization energy is the minimum energy required to remove an electron from a gaseous atom.
Why do cations move toward the cathode?
Because the cathode carries negative charge, attracting positively charged cations.
What are the five common biological cations?
Na⁺, K⁺, Ca²⁺, Mg²⁺, and H⁺.
The Bottom Line
Cations are formed when atoms or molecules lose electrons, resulting in a positive charge. This process typically occurs through oxidation, ionization, or electron transfer reactions. Metals readily form cations due to their low ionization energies and tendency to achieve stable electronic configurations. Cations play critical roles in chemical reactions, electrochemistry, and biological systems.

The Science Editorial Team creates clear, accurate, and student-friendly explanations of scientific concepts across biology, chemistry, physics, taxonomy, anatomy, and related life sciences. Content is written for educational and informational purposes using standard academic references and reliable sources. Read more about the → Science Editorial Team