Quick Answer: A homogeneous mixture has a uniform composition throughout and exists as a single phase, whereas a heterogeneous mixture has a non-uniform composition with two or more visibly distinct phases. The key difference lies in the distribution of components.
This classification is fundamental in analytical chemistry, material science, and laboratory separation techniques.
Understanding mixture classification is a core topic in chemistry when studying the structure and behavior of matter.
Comparison Between Homogeneous and Heterogeneous Mixtures
| Feature | Homogeneous Mixture | Heterogeneous Mixture |
| Definition | Mixture with uniform composition throughout | Mixture with non-uniform composition |
| Phases | Single visible phase | Two or more visible phases |
| Particle Distribution | Evenly distributed | Unevenly distributed |
| Visibility of Components | Not distinguishable by naked eye | Often visibly distinguishable |
| Examples | Saltwater, air, vinegar, alloys | Oil and water, sand in water, soil |
What is a Homogeneous Mixture?
A homogeneous mixture is a mixture in which the components are evenly distributed and cannot be distinguished visually. It appears as a single macroscopic phase, meaning no visible boundaries exist between components.
Key characteristics:
- Uniform composition throughout due to even molecular or ionic dispersion
- Also known as a solution
In a solution, the substance that dissolves is called the solute, while the substance in which it dissolves is known as the solvent. The solvent acts as the dispersion medium that evenly distributes solute particles at the molecular or ionic level.
- Properties are consistent throughout the sample
Examples:
- Salt dissolved in water
- Air (mixture of gases)
- Vinegar (acetic acid in water)
- Brass (alloy of copper and zinc)
What is a Heterogeneous Mixture?
A heterogeneous mixture contains components that are unevenly distributed. The mixture shows more than one visible phase.
Key characteristics:
- Non-uniform composition
- Multiple phases may be present
- Components may be visible to the naked eye
- Particles may settle over time (in suspensions)
Examples:
- Oil and water
- Sand in water
- Soil
- Granite
Core Concept: Phase and Uniformity
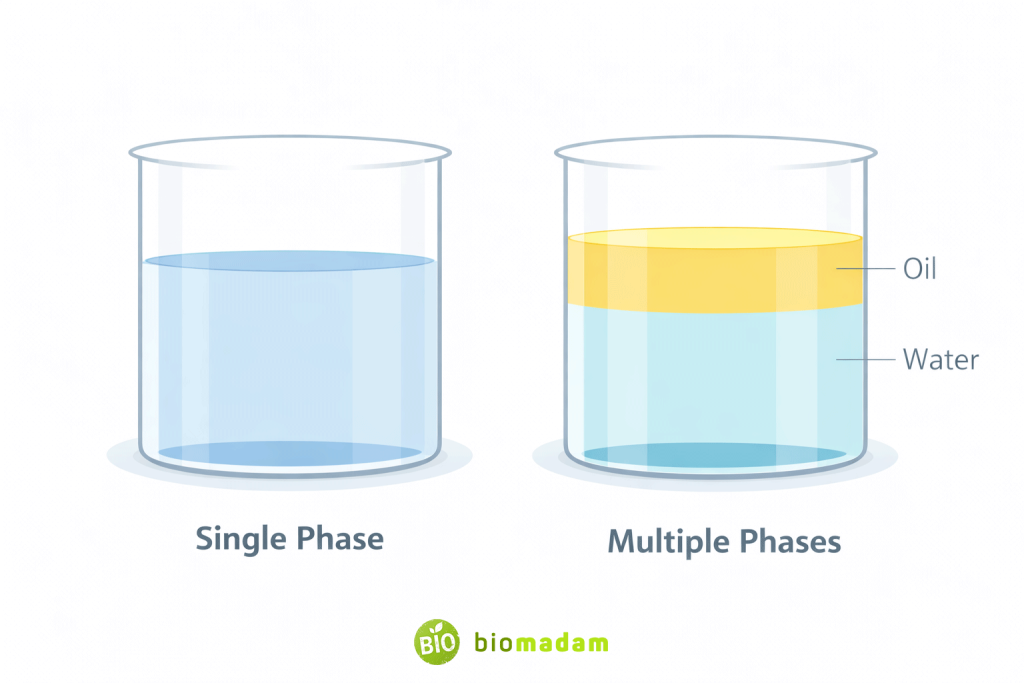
The fundamental difference is based on phase distribution:
- Homogeneous mixtures appear as one phase.
- Heterogeneous mixtures show two or more phases.
A phase is a physically and chemically uniform portion of matter separated by visible or invisible boundaries from other portions.
For example:
- Saltwater appears completely uniform (one phase).
- Oil and water clearly form two layers (two phases).
Types Within Each Category

Homogeneous Mixtures
These are commonly called solutions, where:
- Solute particles are dissolved at molecular or ionic level.
- No settling occurs over time.
Heterogeneous Mixtures
These may include:
- Suspensions (large particles that settle over time)
For a detailed comparison of dispersed systems, review the Difference Between Solution and Suspension.
- Colloids (intermediate-sized particles that do not settle easily but are microscopically heterogeneous)
Colloids exhibit the Tyndall effect, meaning they scatter light due to dispersed particles typically ranging between 1–1000 nanometers in size. This property helps distinguish colloids from true homogeneous solutions.
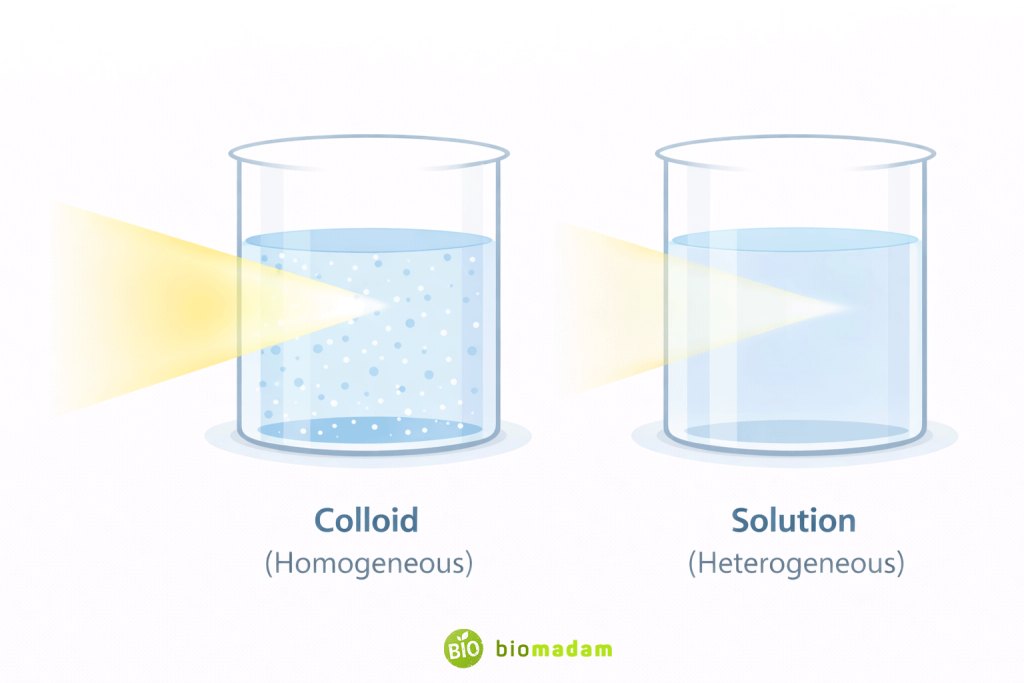
A structured comparison of these systems is explained in the Difference Between Solution, Suspension, and Colloid.
Separation and Physical Behavior
Both homogeneous and heterogeneous mixtures can be separated by physical methods.
This distinction aligns with the Difference Between Physical And Chemical Change, where physical processes do not alter chemical identity.
However, the method depends on the type:
Homogeneous mixtures:
- Distillation
- Evaporation
- Chromatography
Heterogeneous mixtures:
- Filtration
- Decantation
- Magnetic separation
The ability to separate by physical means confirms that both are mixtures rather than pure substances.
To understand how mixtures differ fundamentally from single-component materials, see the Difference Between Pure Substance and Mixture.
How to Identify a Homogeneous or Heterogeneous Mixture
Ask these questions:
- Can you see different parts or layers?
- Does the composition look uniform throughout?
- Does the mixture settle over time?
If it looks uniform and remains consistent, it is likely homogeneous.
If distinct phases or particles are visible, it is heterogeneous.
Is One Type Better Than the Other?
Neither homogeneous nor heterogeneous mixtures are better or worse; they simply describe how substances are distributed within a mixture. The classification depends entirely on uniformity and phase distribution, not quality or usefulness.
Common Confusion Alert
Many students confuse colloids with homogeneous mixtures.
Although colloids may appear uniform to the naked eye, they contain dispersed particles and are technically classified as heterogeneous mixtures.
Key Differences Summary
- Homogeneous mixtures → uniform composition, single phase
- Heterogeneous mixtures → non-uniform composition, multiple phases
- Homogeneous mixtures → components not visibly distinguishable
- Heterogeneous mixtures → components often visibly distinct
- Both → separable by physical methods
Homogeneous or Heterogeneous: 5 Real-Life Examples
- Milk is a colloid and is technically a heterogeneous mixture.
- Fog is a heterogeneous mixture of liquid droplets dispersed in air.
- Saltwater is a homogeneous mixture because salt dissolves completely at the molecular level.
- Granite is heterogeneous due to visibly different mineral phases.
- Air is a homogeneous mixture because its gases (mainly nitrogen and oxygen) are uniformly distributed and exist as a single phase under normal conditions.
FAQs
Is air homogeneous or heterogeneous?
Air is a homogeneous mixture of gases because its components are evenly distributed and exist as a single phase.
Are colloids homogeneous or heterogeneous?
Colloids are technically heterogeneous mixtures, even though they may appear uniform.
Can homogeneous mixtures be separated?
Yes. Homogeneous mixtures can be separated using physical methods such as distillation, evaporation, or chromatography.
Do heterogeneous mixtures always have visible particles?
Often yes, but not always to the naked eye. Some may require magnification to observe dispersed phases clearly.
Is milk homogeneous or heterogeneous?
Milk appears uniform but is actually a colloid. Therefore, it is technically classified as a heterogeneous mixture.
Are all solutions homogeneous?
Yes. By definition, all true solutions are homogeneous mixtures because their solute particles are evenly distributed at the molecular or ionic level.
Can a heterogeneous mixture become homogeneous?
Yes. Some heterogeneous mixtures can become homogeneous if the dispersed components dissolve completely, such as sugar dissolving in water.
What is the main difference in one sentence?
Homogeneous mixtures have uniform composition and a single phase, while heterogeneous mixtures have non-uniform composition and multiple phases.
The Bottom Line
Homogeneous and heterogeneous mixtures differ primarily in uniformity and phase distribution. A homogeneous mixture appears as a single uniform phase, while a heterogeneous mixture contains visibly distinct components or multiple phases. Understanding this distinction helps in classification of matter and selection of appropriate separation techniques.

The Science Editorial Team creates clear, accurate, and student-friendly explanations of scientific concepts across biology, chemistry, physics, taxonomy, anatomy, and related life sciences. Content is written for educational and informational purposes using standard academic references and reliable sources. Read more about the → Science Editorial Team
