Quick Answer: Water is a compound, not a mixture, because hydrogen and oxygen are chemically combined in a fixed ratio (2:1) and cannot be separated by physical methods.
What is Water?
Water is a universal solvent essential for all forms of life. It plays a critical role in biological processes such as digestion, circulation, temperature regulation, and chemical reactions inside cells.
Chemically, a water molecule consists of two hydrogen atoms covalently bonded to one oxygen atom, giving it the molecular formula H₂O.
This fixed composition is the key to understanding whether water is classified as a compound or a mixture.
Water can exist in three physical states—solid (ice), liquid (water), and gas (water vapor)—depending on temperature and pressure. These state changes are physical changes and do not alter the chemical composition of water.
What is a Compound?
A compound is a pure substance formed when two or more different elements combine chemically in a fixed ratio.
Key characteristics of compounds:
- Elements are chemically bonded
- Have a definite composition
- Properties are different from constituent elements
- Cannot be separated by physical methods
- Can only be broken down by chemical reactions
Example: Water (H₂O), sodium chloride (NaCl), carbon dioxide (CO₂)
What is a Mixture?
A mixture is formed when two or more substances are physically combined, not chemically bonded. This distinction is explained in detail in the Difference Between Pure Substance and Mixture.
Key characteristics of mixtures:
- Components retain their original properties
- Composition is variable
- Can be separated by physical methods
- No chemical bonding involved
Example: Salt water, air, soil, sand and iron filings
Is Water a Compound or a Mixture?
Water is clearly a compound.
A water molecule is formed when hydrogen and oxygen chemically combine through covalent bonds in a fixed 2:1 ratio. The resulting substance (water) has properties entirely different from hydrogen or oxygen gases.
Additionally, water cannot be separated into hydrogen and oxygen by physical methods such as filtration or distillation. Chemical methods like electrolysis are required.
According to the law of definite proportions, a compound always contains the same elements in the same ratio by mass. Water strictly follows this law, confirming its classification as a compound.
Scientific Evidence That Water is a Compound
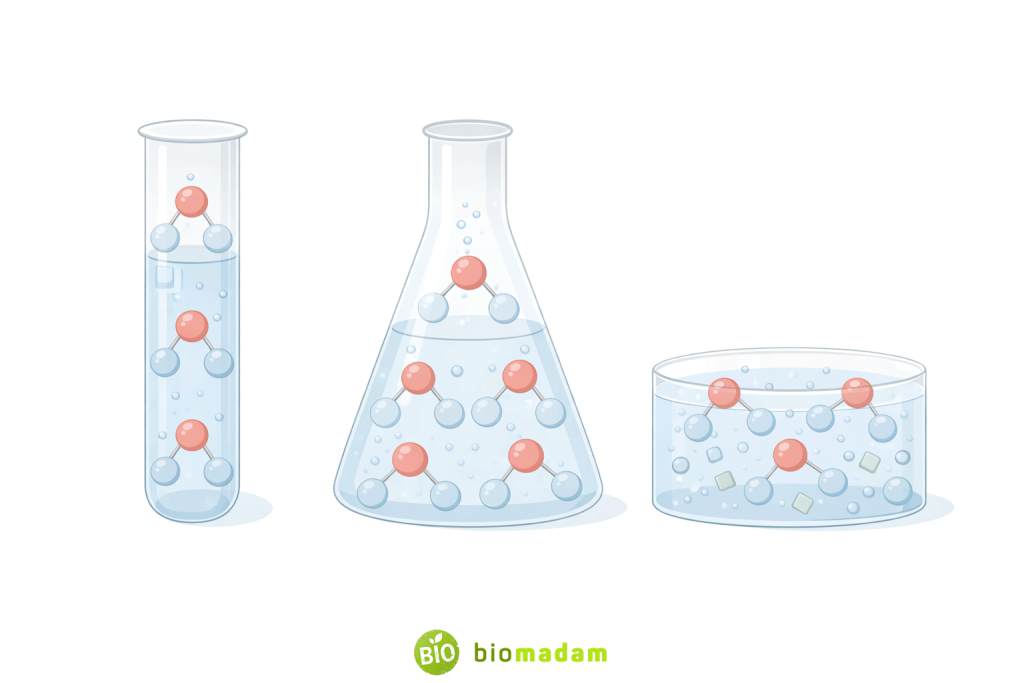
Water is classified as a compound based on established chemical laws and experimental evidence. According to the law of definite proportions, a compound always contains the same elements in a fixed ratio by mass. In water, hydrogen and oxygen are always present in a fixed atomic ratio of 2:1.
This composition can be experimentally verified through electrolysis, a chemical process in which water decomposes into hydrogen and oxygen gases. The gases are released in a consistent 2:1 volume ratio, confirming that water is a chemically bonded substance rather than a physical mixture.
Because its components are chemically combined and can only be separated through chemical reactions, water meets all the criteria of a compound.
Why Water is a Compound (Key Points)
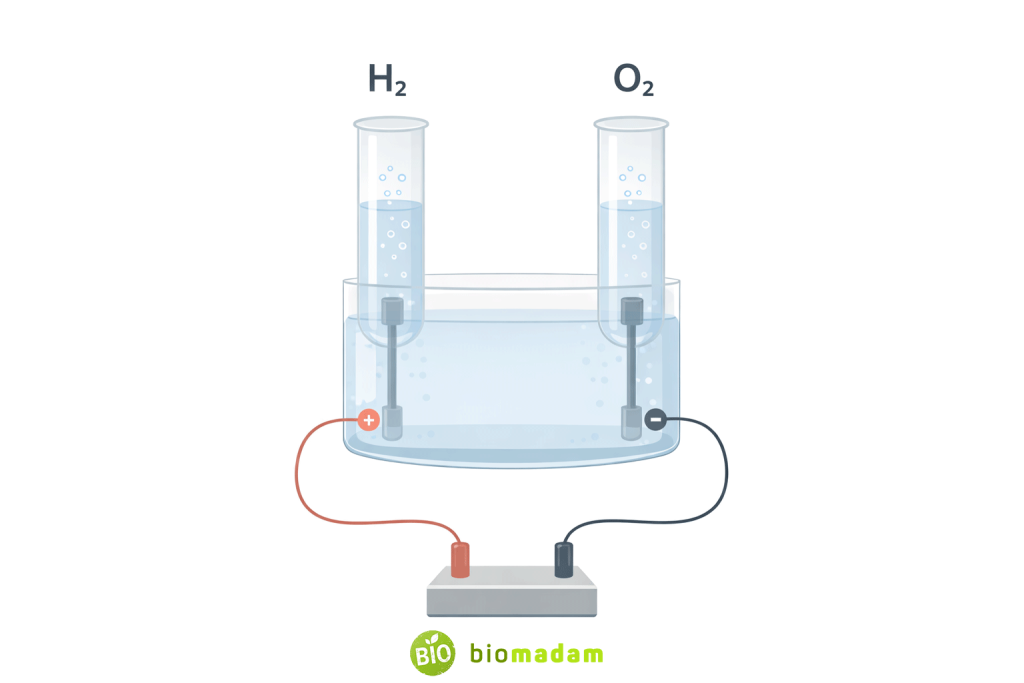
- Contains two different elements
- Elements are chemically bonded
- Has a fixed composition (H₂O)
- Shows new properties
- Obeys the law of definite proportions
- Requires chemical methods for separation
| Feature | Water (Compound) | Mixture |
| Composition | Fixed (H₂O) | Variable |
| Type of bonding | Chemical (covalent) | Physical |
| Properties | New properties formed | Original properties retained |
| Separation method | Chemical (electrolysis) | Physical (filtration, distillation) |
| Example | Distilled water | Salt water |
For a broader classification discussion, see the Difference Between Compounds and Mixtures.
Why Water is Not a Mixture
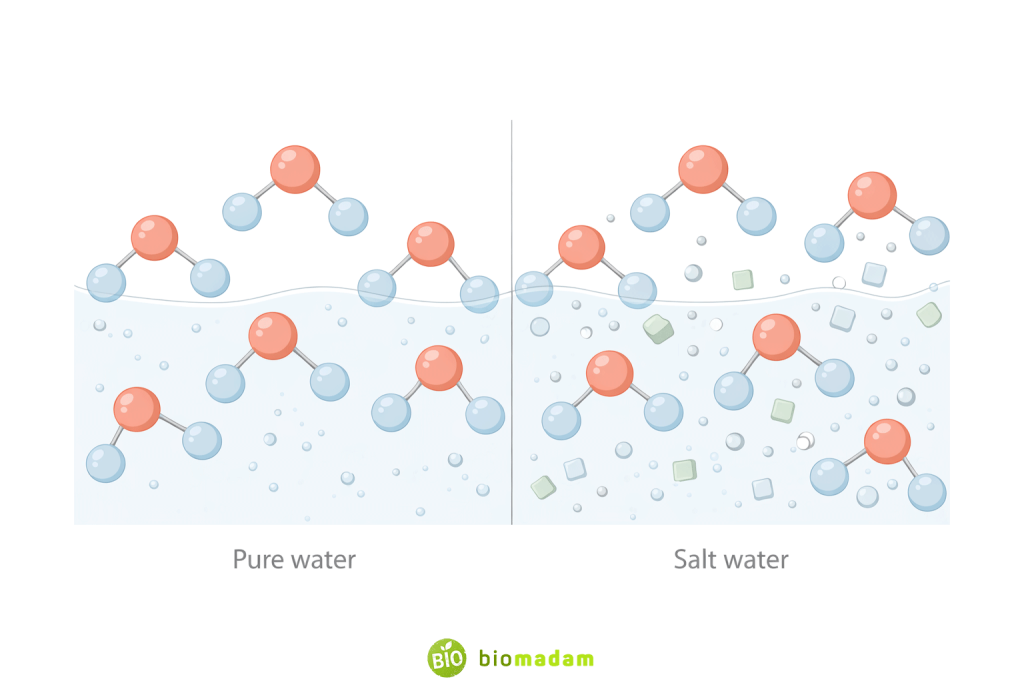
- Mixtures have variable composition, water does not
- Mixtures retain properties of components, water does not
- Mixtures are separated physically, water is not
- No free hydrogen or oxygen exists in water
Pure Water vs Impure Water (Common Confusion)
Pure water refers to chemically pure H₂O, such as distilled or deionized water. In this form, water contains only hydrogen and oxygen atoms chemically bonded in a fixed 2:1 ratio and is therefore classified as a compound.
However, water found in everyday life—such as tap water, river water, mineral water, or seawater—is not chemically pure. These natural sources contain dissolved salts, gases, and minerals. Because additional substances are physically present, they are classified as mixtures, even though the water molecules themselves remain compounds.
Summary
- Pure water (H₂O) → Compound
- Tap water / sea water / mineral water → Mixture
Difference Between Compound and Mixture (Water Example)
| Feature | Water (Compound) | Mixture |
| Composition | Fixed (H₂O) | Variable |
| Bonding | Chemical (covalent) | Physical |
| Properties | New properties | Retained |
| Separation | Chemical methods | Physical methods |
| Example | Pure water | Salt water |
Final Verdict
Water is not a mixture.
It is a compound because hydrogen and oxygen are chemically bonded in a fixed ratio, producing a substance with properties different from its elements.
Understanding this distinction helps students correctly classify substances and strengthens foundational chemistry concepts.
Common Misconceptions About Water
- Ice, liquid water, and steam are different physical states of the same compound (H₂O), not different substances.
- Changing the state of water does not change its chemical composition.
This reflects the concept explained in the Difference Between Physical And Chemical Change.
- Dissolving salt or sugar in water forms a mixture, not a new compound.
- Water cannot be classified as an element because it contains more than one type of atom.
- Understanding these points helps distinguish between physical changes and chemical composition.
FAQs
Is water an element?
No. Water contains two elements, hydrogen and oxygen.
Is water a compound or mixture in daily life?
Pure water is a compound; natural water is usually a mixture.
Can water be separated into hydrogen and oxygen physically?
No. Chemical methods like electrolysis are required.
Why does water obey the law of definite proportions?
Because it always contains hydrogen and oxygen in a fixed ratio.
Is distilled water a compound?
Yes. Distilled water is pure H₂O and therefore a compound.

The Science Editorial Team creates clear, accurate, and student-friendly explanations of scientific concepts across biology, chemistry, physics, taxonomy, anatomy, and related life sciences. Content is written for educational and informational purposes using standard academic references and reliable sources. Read more about the → Science Editorial Team
