Quick Answer: The main difference between X-rays and gamma rays lies in their origin. X-rays are produced by high-energy electron interactions outside the atomic nucleus, while gamma rays are emitted from unstable radioactive nuclei. Gamma rays generally have higher energy and penetration, whereas X-rays are widely used for controlled medical imaging and diagnostics.
Overview
X-rays and gamma rays are both forms of electromagnetic radiation and belong to the high-energy end of the electromagnetic spectrum. Although they share many similarities, they differ in origin, energy range, penetration power, and applications.
Both types are widely used in medicine, research, and industry. Understanding their differences requires knowledge of electromagnetic radiation and nuclear processes studied in physics, particularly in modern and nuclear physics.
What Are X-Rays?
X-rays are high-energy electromagnetic waves produced when fast-moving electrons suddenly decelerate after striking a metal target.
Key Characteristics of X-Rays
- Produced by electron interactions
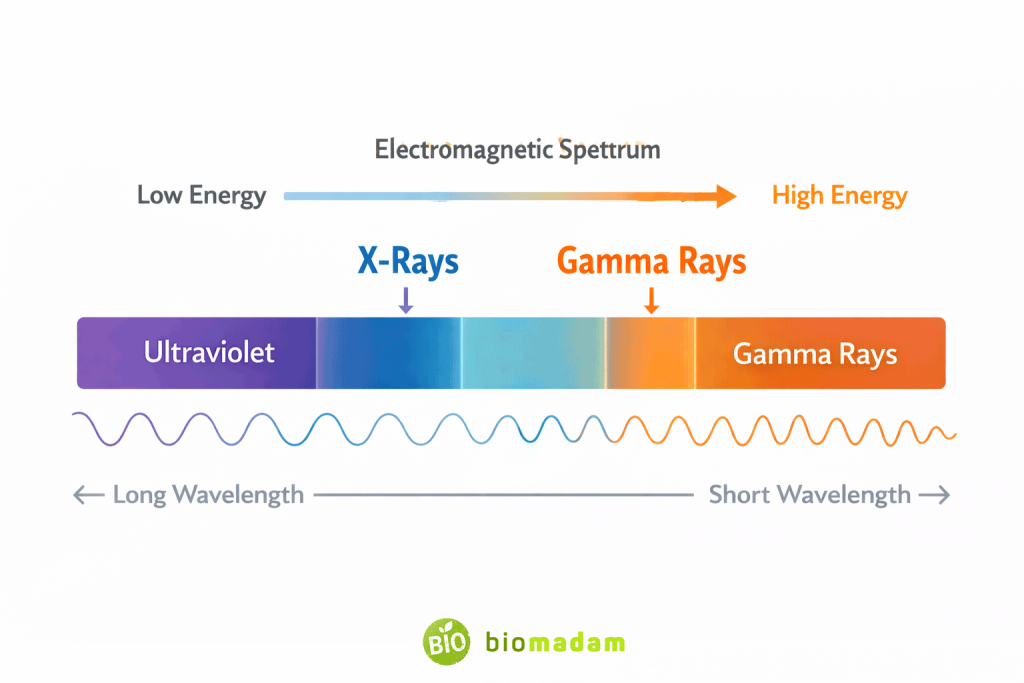
- Wavelength range: ~0.01 to 10 nanometers
- Ionizing radiation
- Moderate to high penetration
- Can travel in vacuum
- No electric charge
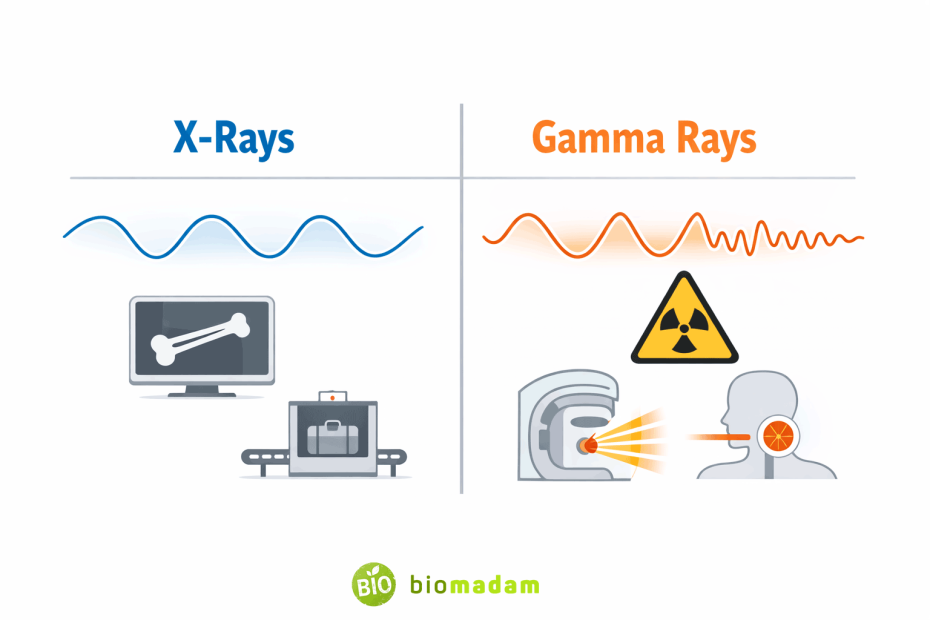
Common Uses of X-Rays
- Medical imaging such as bone fracture detection and dental scans, especially when compared with techniques like X-ray vs MRI that use magnetic fields instead of ionizing radiation.
- Airport and security scanning
- Industrial defect detection
- Astronomy and material analysis
Medical X-rays are considered safe when used in controlled, low doses.
What Are Gamma Rays?
Gamma rays are extremely high-energy electromagnetic waves emitted from the nuclei of radioactive atoms during nuclear transitions.
These emissions commonly occur during processes such as alpha decay and beta decay, where unstable nuclei release energy to become more stable.
Key Characteristics of Gamma Rays
- Originates from radioactive nuclei
- Shorter wavelength than X-rays
- Very high photon energy
- Extremely high penetration
- Ionizing radiation
- No mass or charge
Common Uses of Gamma Rays
- Cancer treatment (radiotherapy)
- Sterilization of medical equipment
- Nuclear medicine
- Industrial radiography
- Astronomical observations
Due to their high energy, gamma rays pose greater biological risks if exposure is uncontrolled.
Comparison Table: X-Rays vs Gamma Rays
| Factor | X-Rays | Gamma Rays |
| Source | Electron interactions | Radioactive nuclei |
| Wavelength | Longer | Shorter |
| Energy | High | Very high |
| Ionization | Ionizing | Ionizing (often stronger) |
| Penetration | High | Very high |
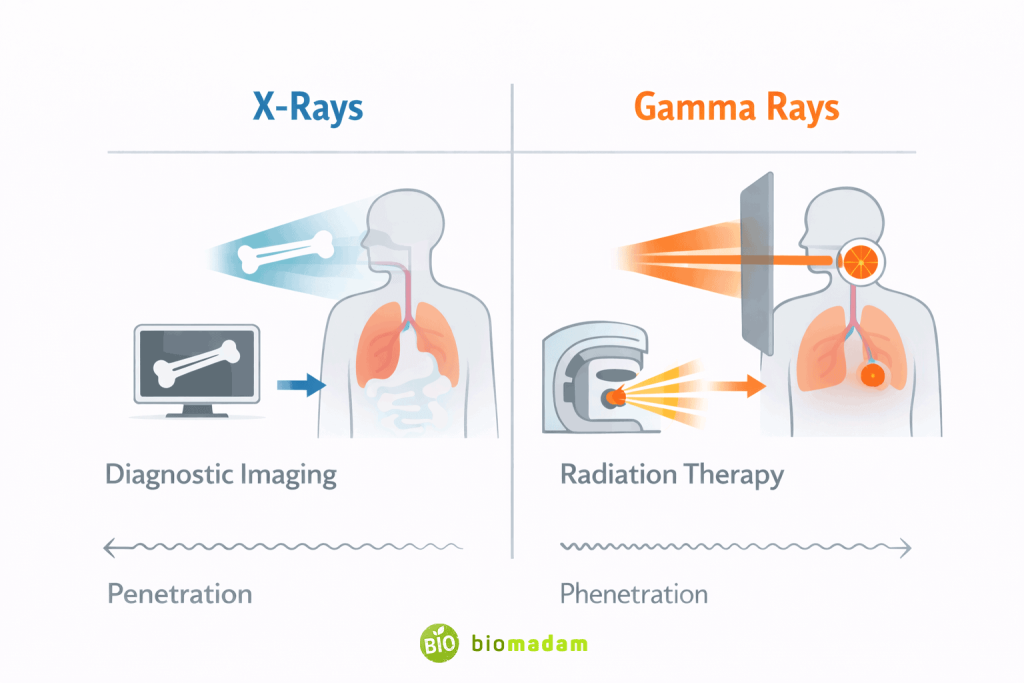
| Medical Use | Diagnostic imaging | Cancer therapy |
| Biological Risk | Dose-dependent | Higher at similar doses |
X-Rays at a Glance
- Produced by electron interactions
- Used mainly for diagnostic imaging
- Moderate to high penetration
- Controlled medical doses are considered safe
Gamma Rays at a Glance
- Emitted from radioactive nuclei
- Used mainly for cancer treatment
- Extremely high penetration
- Higher biological risk if exposure is uncontrolled
Key Differences Between X-Rays and Gamma Rays
1. Source of Origin
- X-rays: Produced outside the nucleus
- Gamma rays: Emitted from unstable atomic nuclei
2. Energy and Penetration
- Gamma rays generally have higher energy and penetration
- X-rays are less penetrating but still effective for imaging
3. Ionizing Ability
- Both are ionizing
- Biological impact depends on energy, dose, and exposure time
4. Medical Application
- X-rays → diagnosis
- In advanced diagnostics, CT scans and MRI are often compared in terms of imaging depth and radiation exposure, as explained in MRI vs CT Scan.
- Gamma rays → treatment
- Since CT technology is based on X-ray imaging, understanding the distinction between CT Scan and X-ray helps clarify their diagnostic capabilities.
Are X-Rays and Gamma Rays Dangerous?
Yes, both are ionizing radiations and can damage biological tissues.
- X-rays: Generally considered safe at low, controlled medical doses
- Gamma rays: Higher risk due to stronger penetration and energy
Medical and industrial use follows strict safety regulations to minimize exposure.
Similarities Between X-Rays and Gamma Rays
- Both are electromagnetic radiation
- Travel at the speed of light
- Consist of photons
- Can ionize matter
- Used in medicine and research
Why Understanding the Difference Matters
- Helps in safe medical application
- Important for physics and radiology exams
- Essential for radiation safety awareness
- Supports accurate scientific classification
The Bottom Line
X-rays and gamma rays are powerful forms of electromagnetic radiation with distinct origins and applications. X-rays are generated by electron interactions and are mainly used for diagnostic imaging, while gamma rays originate from radioactive nuclei and are used primarily in cancer treatment. Although both are ionizing, their energy levels, penetration, and biological effects differ, making proper understanding and controlled use essential.
Frequently Asked Questions (FAQs)
Which is more penetrating: X-rays or gamma rays?
Gamma rays are generally more penetrating due to their higher energy.
Are gamma rays more dangerous than X-rays?
Yes, gamma rays usually pose a higher risk, but actual danger depends on dose and exposure duration.
Can X-rays and gamma rays be distinguished by wavelength alone?
No. Their energy ranges can overlap; origin is the most reliable distinction.
Are both used in medicine?
Yes. X-rays are used for diagnosis, while gamma rays are used for therapy.
References
- https://www.britannica.com/science/X-ray
- https://www.britannica.com/science/gamma-ray
- https://www.nrc.gov/reading-rm/basic-ref/students/science-101/what-is-radiation.html
- https://www.cdc.gov/nceh/radiation
- https://www.nature.com/scitable

The Science Editorial Team creates clear, accurate, and student-friendly explanations of scientific concepts across biology, chemistry, physics, taxonomy, anatomy, and related life sciences. Content is written for educational and informational purposes using standard academic references and reliable sources. Read more about the → Science Editorial Team