Quick Answer: Thin layer chromatography (TLC) is used to separate, identify, and analyze compounds in a mixture. Its major uses include compound detection, identification, purity testing, reaction monitoring, checking column chromatography fractions, and applications in pharmaceuticals, food analysis, forensics, and biochemical research. It is widely used as a rapid, cost-effective analytical technique in laboratory and research settings.
What is Thin Layer Chromatography?
Thin layer chromatography (TLC) is a widely used analytical separation technique in chemistry for mixtures of non-volatile compounds. It works by separating compounds based on their rate of movement over a thin layer of adsorbent material coated on a plate or sheet.
The TLC plate usually consists of:
- Support: glass, plastic, or aluminum foil
- Adsorbent (stationary phase): silica gel, aluminum oxide, or cellulose
The mobile phase (eluent) carries the sample mixture along the plate, allowing separation of components.
Principle of Separation in Thin Layer Chromatography
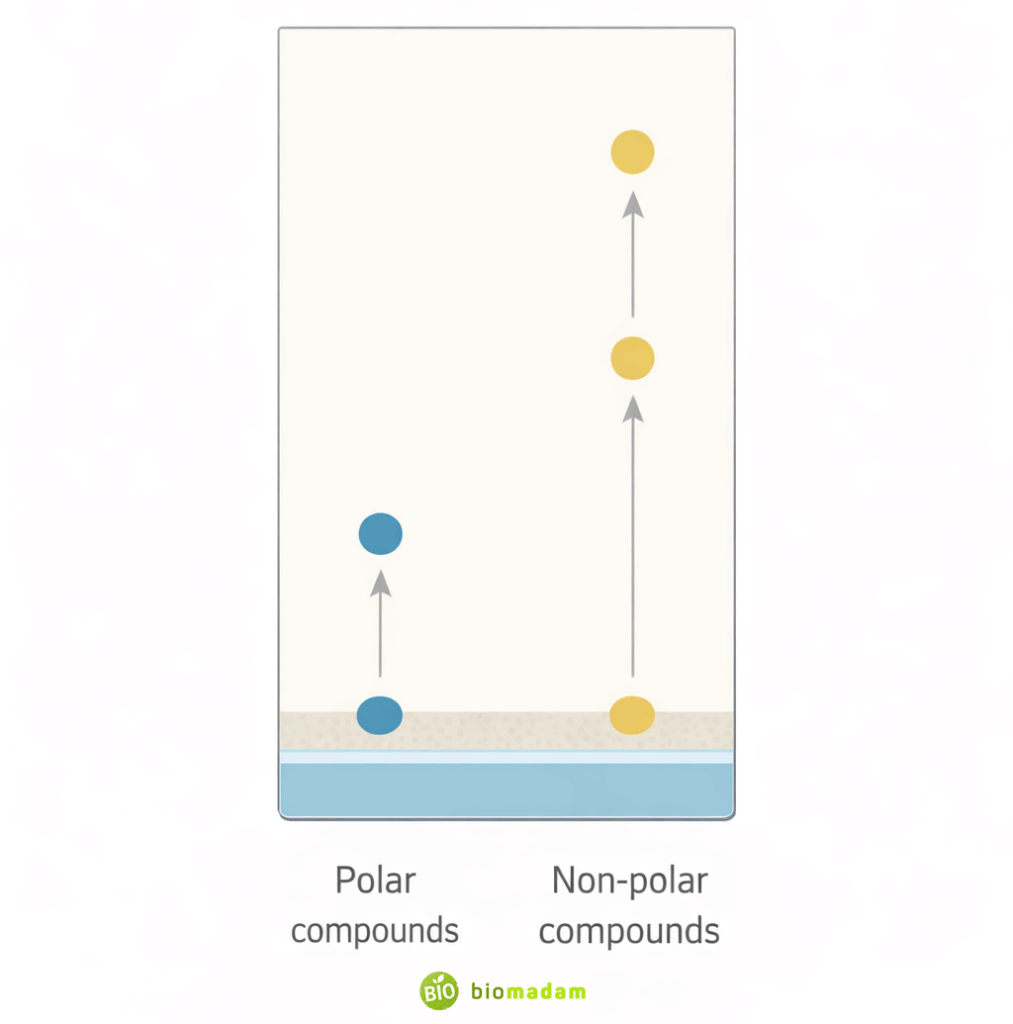
The separation in TLC occurs due to differences in the affinity of compounds for the stationary and mobile phases. The theoretical basis of this separation process is discussed in detail in the Principle of Chromatography.
- Compounds that interact strongly with the stationary phase move slowly.
- Compounds with weaker interactions move faster with the mobile phase.
For example, in normal-phase TLC:
- The stationary phase (silica gel) is polar.
- Polar compounds bind more strongly to silica gel and travel shorter distances.
- Non-polar compounds move farther along the plate.
This difference in movement forms the basis for TLC separation.
The separation in TLC primarily occurs through adsorption, though partition may also contribute depending on the stationary phase used.
Types of Thin Layer Chromatography
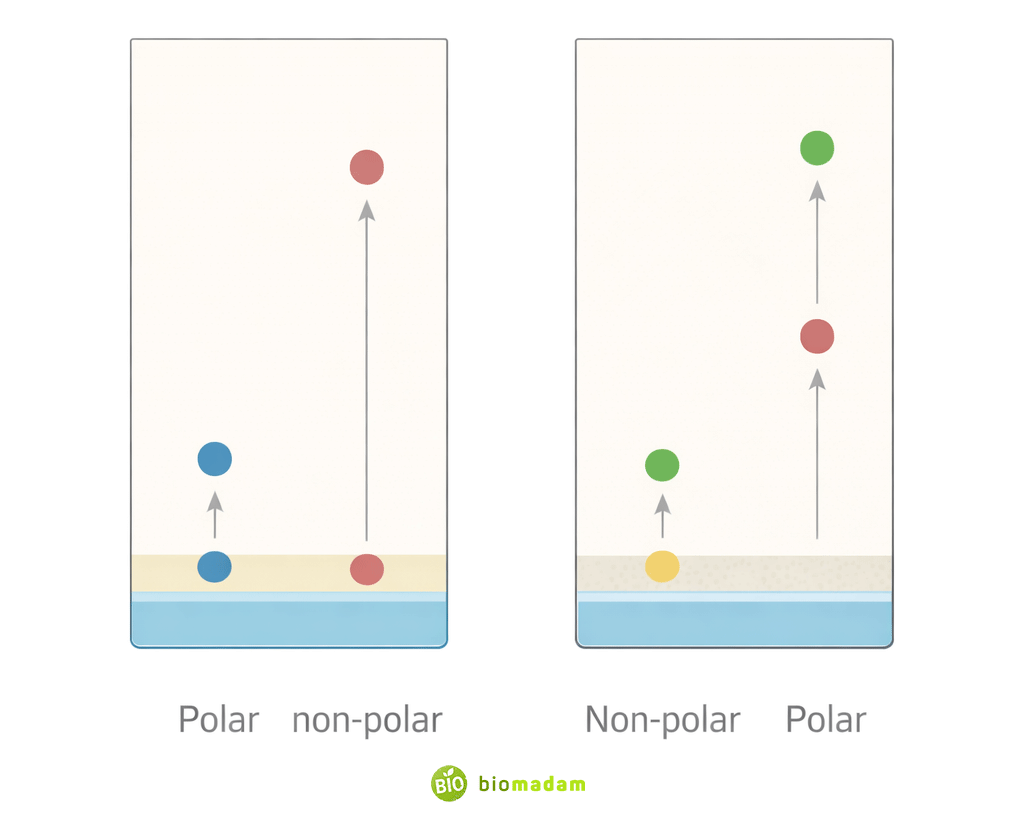
Based on the nature of the stationary phase, thin layer chromatography is mainly classified into two types:
Normal-Phase TLC
In normal-phase TLC, the stationary phase is polar (commonly silica gel or aluminum oxide), while the mobile phase is relatively non-polar. Polar compounds interact strongly with the stationary phase and therefore move slowly, whereas non-polar compounds travel farther.
Reverse-Phase TLC
In reverse-phase TLC, the stationary phase is non-polar and the mobile phase is relatively polar. This type is commonly used in biochemical and pharmaceutical analyses where polar solvents are preferred.
Uses of Thin Layer Chromatography
1. Detection of Compounds in a Mixture
One of the most common uses of TLC is detecting the presence of compounds in a sample.
In many laboratory procedures, extraction results in a mixture of compounds. TLC helps determine whether a compound of interest is present by comparing the sample with a known standard.
Example: To detect phytoestrogens in a plant extract, the extract is run alongside a pure phytoestrogen standard. If both produce spots at the same position, the compound is likely present.
2. Identification of Compounds Using Rf Values
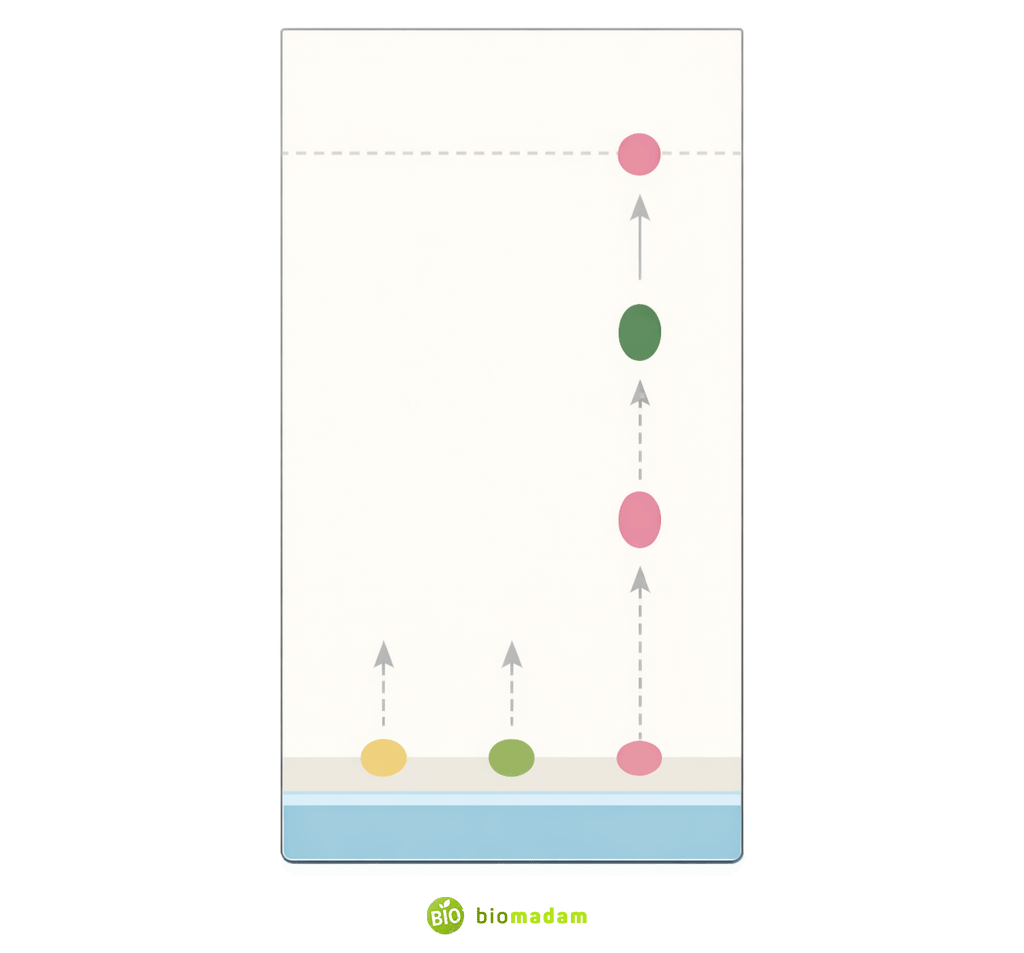
Thin layer chromatography is extensively used for compound identification.
Each compound has a characteristic Rf value (retention factor) under fixed experimental conditions. When two samples show identical Rf values, it suggests they are the same compound.
This method allows identification of compounds:
- From different sources
- In unknown mixtures
- During comparative analysis
Exam Tip: Identical Rf values under the same conditions indicate identical compounds.
Rf Value Formula
The retention factor (Rf) is calculated using the following formula:
Rf = Distance traveled by the compound ÷ Distance traveled by the solvent front
The Rf value is always less than 1 and remains constant for a compound under identical experimental conditions.
3. Determining the Number of Compounds in a Mixture
TLC is a simple and effective method to determine how many components are present in a mixture.
- Each distinct spot corresponds to a different compound.
- The number of visible spots indicates the minimum number of components.
This application is especially useful in:
- Preliminary analysis
- Natural product research
- Crude extract screening
4. Monitoring the Progress of Chemical Reactions
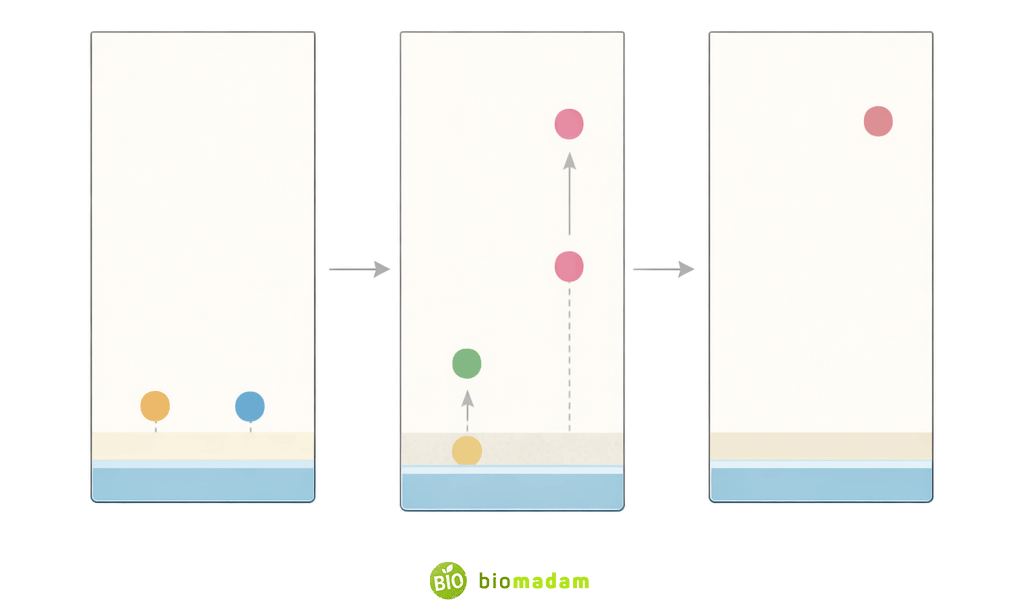
Thin layer chromatography is widely used to monitor chemical reactions over time.
Reaction samples are collected at different time intervals and analyzed using TLC to observe:
- Disappearance of reactants
- Appearance of products
By comparing TLC plates at different time points, researchers can assess reaction completion and rate.
This application is common in:
- Organic synthesis
- Pharmaceutical chemistry
- Research laboratories
Real-World Application Example: In pharmaceutical synthesis, thin layer chromatography is routinely used to monitor reaction progress during drug development. Chemists analyze reaction mixtures at regular intervals to ensure complete conversion of reactants into the desired product before proceeding to purification.
5. Determining the Purity of Compounds
TLC is frequently used to assess compound purity.
- A single spot indicates a relatively pure compound.
- Multiple spots suggest impurities or incomplete purification.
This use is critical in:
- Purification experiments
- Drug synthesis
- Food quality analysis
Example: After purification, a compound is run alongside a standard. If only one spot appears at the same position as the standard, the purification is considered successful.
6. Monitoring Column Chromatography Separations
Thin layer chromatography is commonly used alongside column chromatography, including methods explained in Types of Column Chromatography.
During column chromatography:
- Multiple fractions are collected.
- TLC helps identify which fractions contain the desired compound.
Fractions showing identical TLC patterns are combined, improving efficiency and accuracy of purification.
Thin layer chromatography is often used alongside other chromatographic techniques, such as column chromatography and paper chromatography, to improve analytical accuracy.
7. Applications in Pharmaceutical Analysis
In pharmaceutical laboratories, TLC is used to:
- Check drug purity
This application is closely related to analytical methods discussed in Application of Thin Layer Chromatography.
- Identify active pharmaceutical ingredients
- Detect degradation products
- Perform quality control tests
Although advanced techniques exist, TLC remains valuable due to its low cost, speed, and simplicity.
8. Applications in Food and Forensic Analysis
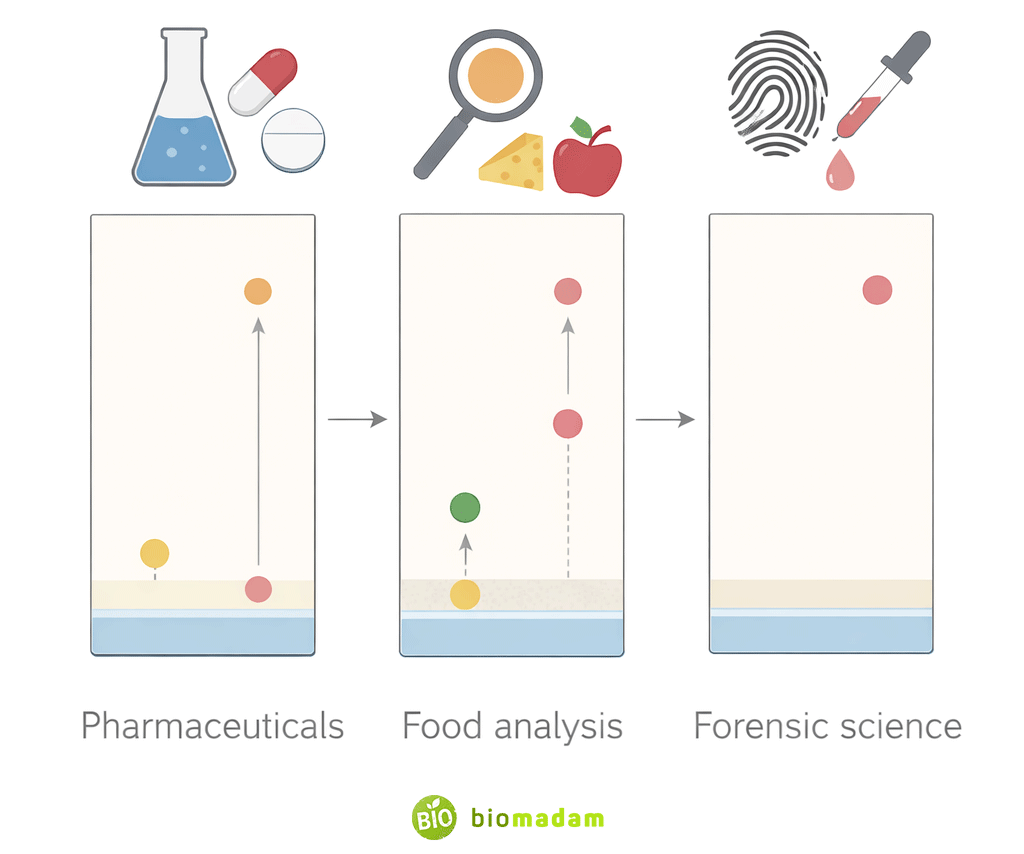
Thin layer chromatography is also applied in:
- Food analysis: detecting adulterants, dyes, and contaminants
- Forensic science: analyzing inks, dyes, drugs, and poisons
Its ability to rapidly separate compounds makes it suitable for screening-level investigations.
Advantages of Thin Layer Chromatography
- Simple and rapid technique
- Requires small sample quantities
- Cost-effective
- Multiple samples analyzed simultaneously
- Visual and easy to interpret
- Allows rapid comparison of multiple samples on a single plate
Limitations of Thin Layer Chromatography
Despite its wide applications, thin layer chromatography has certain limitations:
- Limited resolution for very complex mixtures
- Mostly qualitative or semi-quantitative in nature
- Results are sensitive to solvent composition and environmental conditions
- Not suitable for highly volatile compounds
Due to these limitations, TLC is often used as a preliminary or supporting analytical technique rather than a standalone method.
A broader evaluation of strengths and weaknesses is discussed in Advantages and Disadvantages of Thin Layer Chromatography.
Conclusion
Thin layer chromatography is a versatile and powerful analytical technique with applications across chemistry, biology, pharmaceuticals, food science, and forensic analysis. Its ability to separate, identify, and analyze compounds quickly makes it an essential tool in both research and routine laboratory work.
FAQs
What is thin layer chromatography mainly used for?
TLC is mainly used to separate, identify, and analyze compounds in a mixture.
How does TLC identify compounds?
By comparing Rf values of unknown samples with known standards.
Can TLC determine purity of a compound?
Yes, a single spot usually indicates purity, while multiple spots suggest impurities.
Why is TLC used in reaction monitoring?
It helps track reactant disappearance and product formation over time.
Is TLC used in pharmaceuticals?
Yes, it is widely used for drug analysis, purity testing, and quality control.
What does Rf value indicate in TLC?
It indicates the relative movement of a compound compared to the solvent front and helps in compound identification.

The Science Editorial Team creates clear, accurate, and student-friendly explanations of scientific concepts across biology, chemistry, physics, taxonomy, anatomy, and related life sciences. Content is written for educational and informational purposes using standard academic references and reliable sources. Read more about the → Science Editorial Team
