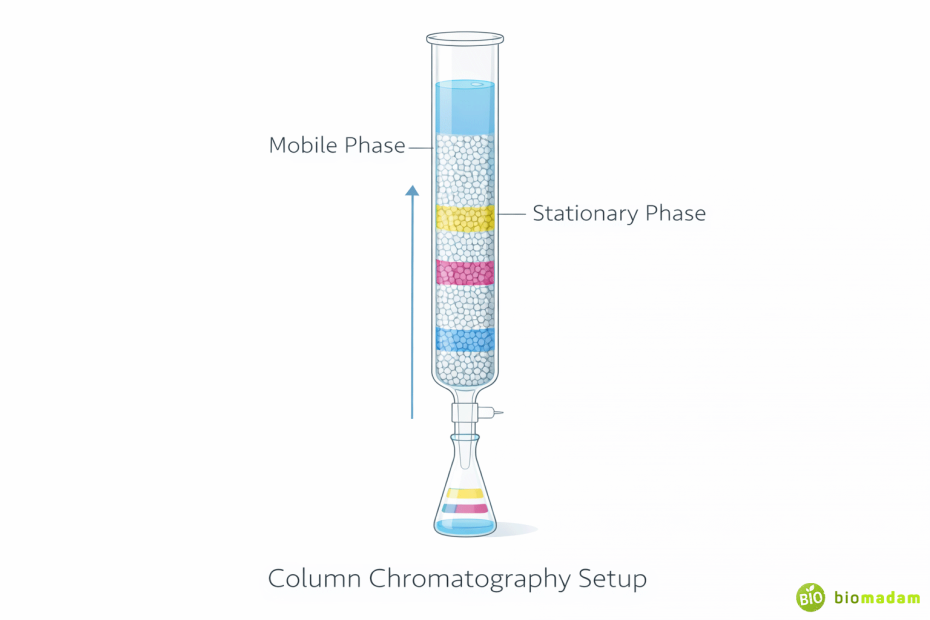
Quick Answer: Column chromatography is a chemistry based separation technique that isolates components of a mixture using a stationary phase packed inside a column and a flowing mobile phase.
Understanding the different types of column chromatography helps students and researchers choose the most suitable method for separating complex mixtures.
The main types of column chromatography include adsorption, ion exchange, gel filtration, affinity, gas chromatography, partition chromatography, and high-performance liquid chromatography (HPLC).
What This Article Covers
- Principle of column chromatography
- Seven major types explained
- How each type works
- Practical examples
- Industrial and research applications
- Comparison of different types
The general separation mechanism is explained in Principle of Chromatography.
Core Concept: Principle of Column Chromatography
Column chromatography separates compounds based on their interaction with:
- Stationary phase (solid or liquid packed inside a column)
- Mobile phase (liquid or gas flowing through the column)
This technique is widely used in organic chemistry, biochemistry, and pharmaceutical research for purification and analysis.
As the mobile phase moves, components of the mixture travel at different speeds depending on:
- Polarity
- Molecular size
- Charge
- Specific binding interactions
This difference causes separation.
The rate at which a compound moves through the column is called its retention time. Compounds with stronger interactions with the stationary phase have longer retention times, while weakly interacting compounds elute faster.
How Are Types of Column Chromatography Classified?
Column chromatography is not classified randomly. The different types are grouped based on the principle of separation.
Some methods separate compounds based on surface adsorption, others on charge, size, solubility, or specific biological interactions.
In many textbooks, certain techniques are considered major categories, while others are treated as specialized subtypes derived from them. Understanding this classification helps avoid confusion when different sources list slightly different numbers of types.
Why Column Chromatography Is Important
Column chromatography is widely used because it:
- Separates complex mixtures
- Purifies biomolecules
- Removes impurities
- Supports pharmaceutical manufacturing
- Enables biochemical research
It remains one of the most versatile laboratory separation techniques.
To understand how column chromatography differs from other separation techniques, it is helpful to compare it with thin layer chromatography and paper chromatography.
Major Types of Column Chromatography
Adsorption Chromatography
Principle: Separation based on adsorption of molecules onto a solid surface.
- Stationary phase: Silica gel or alumina
- Mobile phase: Liquid solvent
Compounds bind to the solid surface via Van der Waals forces and steric interactions.
Example: Separation of plant pigments (chlorophyll and carotenoids).
This was first demonstrated by Mikhail Tsvet in 1901.
In simple terms, the more strongly a compound sticks to the solid surface, the slower it moves through the column.
Ion Exchange Chromatography
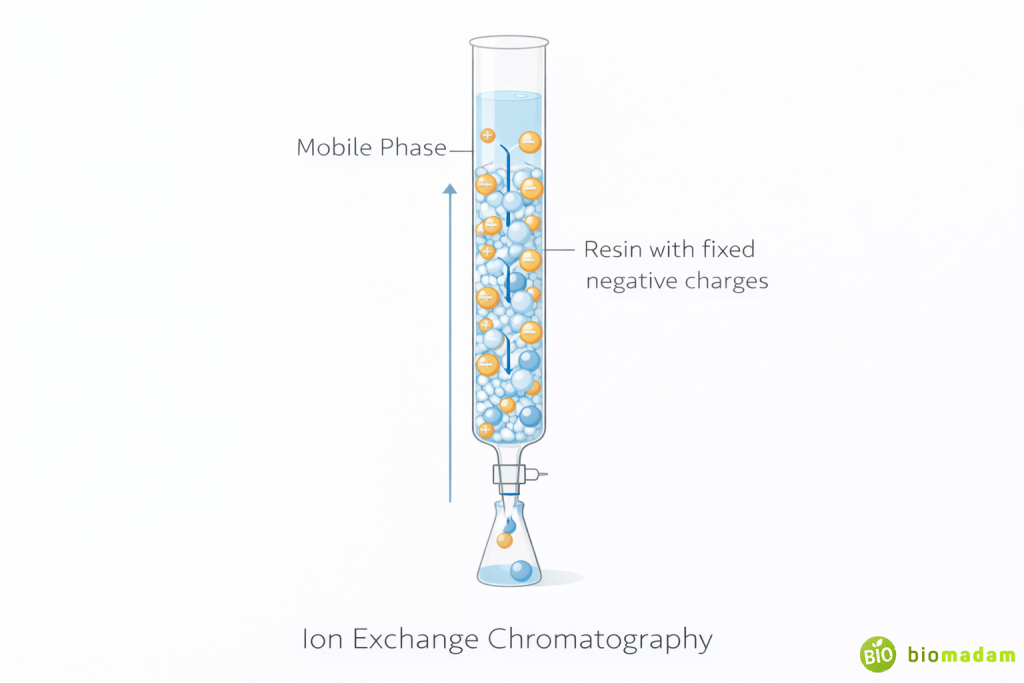
Principle: Separation based on charge differences.
- Stationary phase: Charged resin
- Molecules bind to opposite charges
Types:
- Cation exchange (for positively charged molecules)
- Anion exchange (for negatively charged molecules)
Applications:
- Protein purification
- Nucleic acid separation
The strength of binding depends largely on pH and ionic strength, which means separation conditions must be carefully controlled.
Practical limitations of this method are discussed in Disadvantages of ION Exchange Chromatography.
Gel Filtration Chromatography (Size Exclusion)
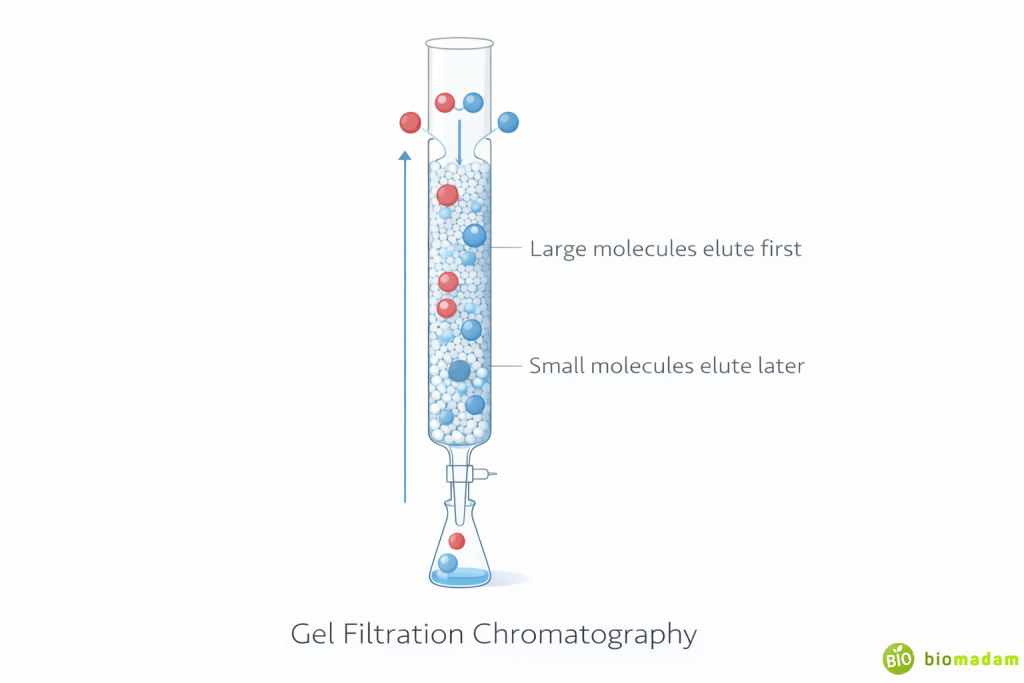
Principle: Separation based on molecular size.
- Larger molecules elute first
- Smaller molecules enter pores and elute later
Stationary phase consists of porous beads forming a molecular sieve.
Applications:
- Protein purification
- Polymer separation
- Macromolecular analysis
Unlike adsorption or ion exchange methods, this technique does not rely on chemical binding. The separation is purely physical.
Affinity Chromatography
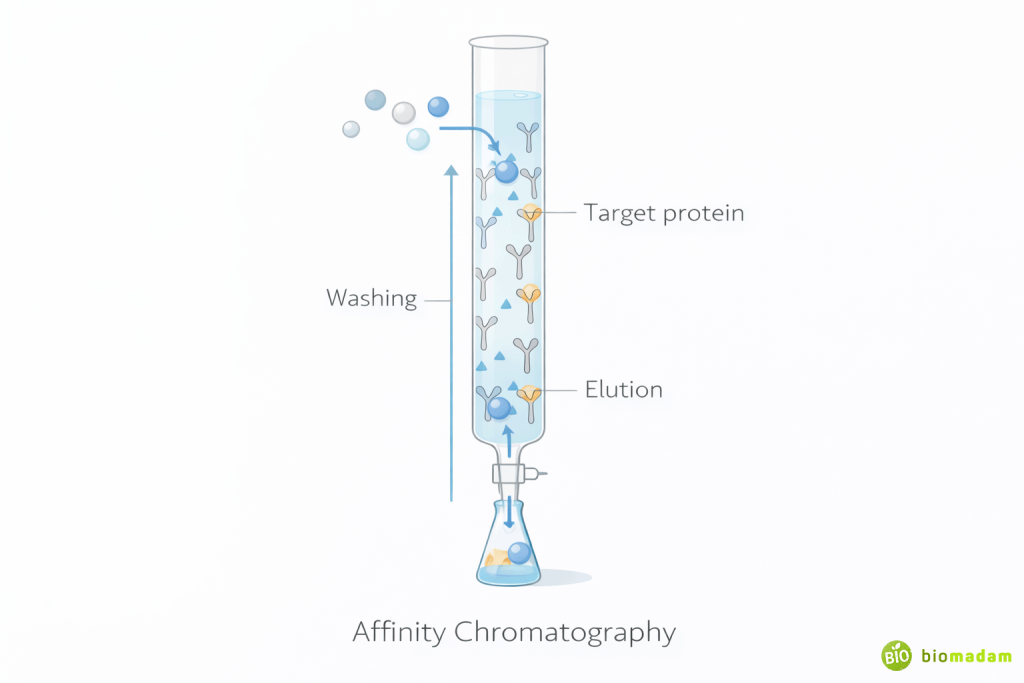
Principle: Separation based on specific biological interactions.
Example:
- Antigen–antibody
- Enzyme–substrate
- Receptor–ligand
Only molecules with specific affinity bind to the stationary phase.
Applications:
- Recombinant protein purification
- Biotechnology research
Because of this highly specific interaction, affinity chromatography often produces very pure results in a single step.
For a balanced understanding, check Disadvantages of Affinity Chromatography.
Gas Chromatography (GC)
Principle: Separation of volatile compounds in gaseous form.
- Mobile phase: Inert gas
- Stationary phase: Liquid/polymer coating inside column
Used for analyzing vapor-phase compounds.
Applications:
- Forensic analysis
- Environmental monitoring
- Industrial quality control
Partition Chromatography
Principle: Separation based on distribution between two liquid phases.
Components distribute differently depending on their partition coefficient.
Applications:
- Lipid analysis
- Pigment separation
- Pharmaceutical purification
High-Performance Liquid Chromatography (HPLC)
Principle: Advanced liquid chromatography using high pressure.
- High-pressure pumps typically operate between 50 and 400 bar, forcing solvent through tightly packed stationary particles for improved separation efficiency.
- Fine stationary particles
- High precision separation
Applications:
- Pharmaceutical manufacturing
- Drug analysis
- Forensic research
- Clinical diagnostics
HPLC provides higher sensitivity and accuracy than traditional column chromatography.
The use of smaller particle sizes and high pressure significantly improves resolution compared to traditional gravity-based columns.
Additional and Specialized Types of Column Chromatography
Beyond these seven primary categories, modern chromatography includes several specialized variations designed for specific research needs.
Reverse Phase Chromatography
Reverse phase chromatography is a subtype of liquid chromatography in which the stationary phase is non-polar and the mobile phase is relatively polar. It is widely used in pharmaceutical analysis because many drug molecules are moderately non-polar.
Normal Phase Chromatography
Normal phase chromatography uses a polar stationary phase (such as silica) and a non-polar solvent. It is often considered a variation of adsorption chromatography.
Flash Column Chromatography
Flash chromatography is an improved version of traditional column chromatography. It uses pressurized gas to push the solvent through the column, making the process faster and more efficient. It is common in organic synthesis laboratories.
Hydrophobic Interaction Chromatography (HIC)
Hydrophobic interaction chromatography is mainly used in protein purification. It separates molecules based on hydrophobic interactions under high-salt conditions.
Chiral Chromatography
Chiral chromatography is designed to separate enantiomers — molecules that are mirror images of each other. This technique is especially important in pharmaceutical research.
Comparison of Different Types
The table below summarizes the major types of column chromatography and their primary separation principles:
| Type | Basis of Separation | Best For |
| Adsorption | Surface interaction | Pigments, small molecules |
| Ion Exchange | Charge | Proteins, nucleic acids |
| Gel Filtration | Size | Macromolecules |
| Affinity | Specific binding | Target biomolecules |
| Gas Chromatography | Volatility | Gaseous compounds |
| Partition | Solubility | Lipids, drugs |
| HPLC | High-pressure precision | Pharmaceutical analysis |
Choosing the Right Type of Column Chromatography
| Situation | Recommended Type |
| Charged biomolecules | Ion exchange |
| Large proteins or polymers | Gel filtration |
| Highly specific purification | Affinity |
| Volatile compounds | Gas chromatography |
| High-precision pharmaceutical analysis | HPLC |
When to Use Each Type
- Use adsorption chromatography for simple separations.
- Use ion exchange for charged biomolecules.
- Use gel filtration for size-based purification.
- Use affinity chromatography for highly specific purification.
- Use GC for volatile compounds.
- Use HPLC when high accuracy and reproducibility are required.
Advantages of Column Chromatography
- High separation efficiency
- Flexible stationary and mobile phases
- Applicable to small and large molecules
- Scalable for industrial production
Limitations
- Time-consuming (traditional columns)
- Requires solvent optimization
- Some techniques require expensive equipment (HPLC)
One-Line Summary
Column chromatography is a versatile separation technique with seven major types that isolate compounds based on polarity, size, charge, or specific binding interactions.
FAQs
1. How many types of column chromatography are there?
There are seven major types: adsorption, ion exchange, gel filtration, affinity, gas chromatography, partition chromatography, and HPLC.
2. Which type is best for protein purification?
Affinity chromatography and ion exchange chromatography are commonly used.
3. What is the difference between HPLC and regular column chromatography?
HPLC uses high pressure for faster and more precise separation.
4. Is gas chromatography a type of column chromatography?
Yes, it uses a column but operates in gaseous mobile phase.
5. Why is column chromatography important in pharmaceuticals?
It helps purify drugs and remove impurities during manufacturing.
6. What are the four main types of column chromatography?
The most commonly recognized main types are adsorption chromatography, ion exchange chromatography, gel filtration (size exclusion) chromatography, and affinity chromatography.
The Bottom Line
The choice among different types of column chromatography depends entirely on the chemical properties of the compound being separated. No single technique is universally superior. Instead, chemists select the method that best matches the molecule’s size, charge, polarity, volatility, or biological specificity.

The Science Editorial Team creates clear, accurate, and student-friendly explanations of scientific concepts across biology, chemistry, physics, taxonomy, anatomy, and related life sciences. Content is written for educational and informational purposes using standard academic references and reliable sources. Read more about the → Science Editorial Team