Quick Answer: Chromatography is an analytical technique used to separate, identify, and analyze components of a mixture by distributing them between a stationary phase and a mobile phase.
Components move at different speeds depending on their chemical affinity, allowing effective separation and analysis in chemistry, biology, pharmaceuticals, and forensic science.
What Is Chromatography?
Chromatography is a fundamental analytical technique widely used in Chemistry and scientific laboratories to separate complex mixtures into individual components. It plays a critical role in chemistry, biology, pharmaceuticals, environmental science, and forensic analysis. The technique allows scientists to separate, identify, and purify compounds based on their chemical behavior and interaction with different phases.
The term chromatography originates from the Greek words chroma (color) and graphein (to write), meaning “color writing.” Although originally developed to separate colored substances, modern chromatography is extensively used for both colored and colorless compounds.
Definition of Chromatography
Chromatography is defined as:
An analytical separation technique in which components of a mixture are distributed between a stationary phase and a mobile phase based on their relative affinities.
This differential distribution enables effective separation, identification, and quantitative analysis of substances.
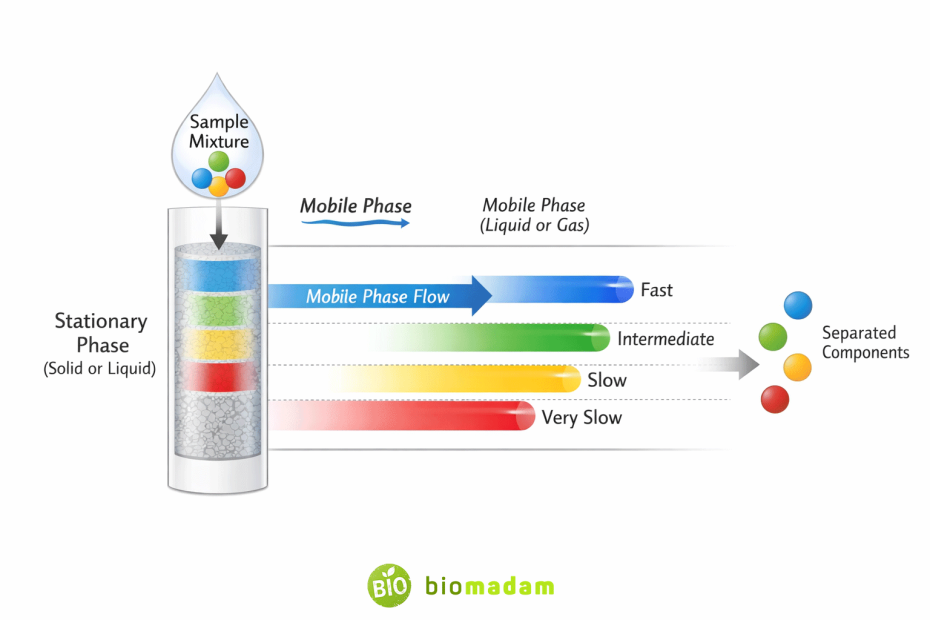
The principle of chromatography is based on the differential distribution of mixture components between a stationary phase and a mobile phase. Components with stronger attraction to the stationary phase move slowly, while those with greater affinity for the mobile phase move faster. This difference in movement results in separation.
Who Introduced Chromatography?
Chromatography was first introduced in 1906 by the Russian botanist Mikhail Tswett. While studying plant pigments such as chlorophyll, carotenoids, and xanthophylls, Tswett used adsorption columns packed with calcium carbonate to separate these pigments. His pioneering work laid the foundation for modern chromatographic techniques used today.
Tswett’s early work was initially overlooked but later gained recognition as chromatography evolved into a cornerstone technique of analytical chemistry. Today, his principles form the basis of advanced systems such as HPLC and GC.
Phases of Chromatography

Chromatography operates using two essential phases:
Stationary Phase
The stationary phase is a solid or liquid that remains fixed during the chromatographic process. It provides a surface on which components of the mixture interact and are temporarily retained.
- It determines selectivity and separation efficiency
- Often porous to increase surface area
- Choice depends on the type of chromatography
Common examples: Silica gel, alumina, cellulose paper, polymer resins
Mobile Phase
The mobile phase is a liquid or gas that moves continuously through the stationary phase, carrying the mixture components along with it.
- Acts as a solvent
- Enables movement and separation of compounds
- Its polarity strongly influences separation
Common examples: Water, ethanol, methanol, acetone, acetic acid, carrier gases (in gas chromatography)
How Does Chromatography Work?
Chromatography works on the principle of differential affinity. When a sample (solute) is introduced into the system, its components distribute themselves between the stationary and mobile phases.
This distribution is governed by the distribution coefficient (K):
- Components with higher affinity for the stationary phase move more slowly
- Components with higher affinity for the mobile phase move faster
As the mobile phase moves, components separate due to differences in their migration rates.
Key Separation Mechanisms
- Adsorption: Interaction between solute and solid stationary phase
- Partition: Distribution of solute between two liquid phases
- Polarity differences: Variation in molecular interactions
- Molecular size and weight: Especially important in size exclusion chromatography
Why Components Separate at Different Speeds
Each compound interacts differently with the stationary and mobile phases due to variations in polarity, solubility, molecular size, and chemical structure. These differences cause components to migrate at distinct rates, resulting in separation.
Factors Affecting Chromatographic Separation
Chromatographic efficiency depends on several factors, including the nature of the stationary phase, composition of the mobile phase, temperature, flow rate, and sample concentration. Optimizing these parameters improves resolution and accuracy
For example, increasing mobile phase polarity can speed up the movement of polar compounds, while changes in temperature can significantly affect gas chromatography efficiency.
Types of Chromatography

Chromatography exists in several forms, each designed for specific analytical needs. These techniques can be broadly classified based on the physical state of the mobile phase, separation mechanism, and application area.
- Gas Chromatography (GC)
- Liquid Chromatography (LC)
- High-Performance Liquid Chromatography (HPLC)
- Thin Layer Chromatography (TLC)
- Paper Chromatography
- Column Chromatography
- Ion Exchange Chromatography
- Size Exclusion Chromatography
- Affinity Chromatography
- Reverse-Phase Chromatography
- Hydrophobic Interaction Chromatography
- Flash Chromatography
Each type differs in stationary phase, mobile phase, and separation mechanism.
Why Chromatography Is Important
Chromatography is essential because many natural and synthetic substances exist as complex mixtures. Without effective separation techniques, accurate identification, purification, and quality control would not be possible in scientific research, medicine, and industry.
Applications of Chromatography

Chromatography is widely applied across multiple scientific and industrial fields.
Food Industry
- Detection of food additives and contaminants
- Quality control and food safety testing
Chemical Industry
- Air and water quality analysis
- Identification of chemical compounds using gas chromatography
Pharmaceutical Industry
- Drug development and purification (Thin layer chromatography applications are explained in detail in Application of Thin Layer Chromatography.)
- Detection of trace impurities
- Quality assurance of pharmaceutical products
Molecular Biology and Biochemistry
- Protein, enzyme, and vitamin separation
- Plasma fractionation and insulin purification using HPLC
- Support for advanced hyphenated techniques
Forensic Science
- Analysis of blood, hair, and tissue samples
- Toxicology and drug identification
- Crime investigation support
Environmental Science
- Detection of pollutants in air, soil, and water
- Monitoring pesticide and heavy metal contamination
Clinical and Medical Diagnostics
- Analysis of blood, urine, and hormones
- Disease marker detection and metabolic studies
Biotechnology and Genetic Research
- Purification of DNA, RNA, and antibodies
- Protein characterization and vaccine development
Petrochemical and Energy Industry
- Analysis of crude oil components
- Fuel quality testing and hydrocarbon profiling
Cosmetics and Consumer Products
- Quality testing of fragrances and skincare products
- Detection of allergens and harmful substances
Advantages of Chromatography
- High separation efficiency
- Applicable to complex mixtures
- High sensitivity and accuracy
- Suitable for both qualitative and quantitative analysis

The strengths and limitations of specific techniques such as TLC are discussed in Advantages and Disadvantages of Thin Layer Chromatography.
Disadvantages of Chromatography
- High initial equipment cost (especially HPLC and GC)
- Requires skilled personnel for accurate operation
- Time-consuming for complex sample preparation
- Some techniques consume large volumes of solvents
- Limited suitability for thermally unstable compounds (in GC)
Despite these limitations, selecting the appropriate chromatographic technique and conditions can minimize drawbacks and maximize analytical efficiency.
Conclusion
Chromatography is an indispensable tool in modern analytical science. Its ability to separate, identify, and purify complex mixtures has revolutionized research and industrial processes across multiple disciplines. With continuous advancements in chromatographic columns, detectors, and analytical software, chromatography will remain a cornerstone technique in scientific discovery and quality control.
Frequently Asked Questions (FAQs)
What is the basic principle of chromatography?
Chromatography works on the principle of differential distribution of mixture components between a stationary phase and a mobile phase based on their chemical affinities.
Why is chromatography important in laboratories?
It allows accurate separation, identification, and quantification of compounds, making it essential in research, diagnostics, and quality control.
What are the two main phases of chromatography?
The two main phases are the stationary phase (fixed) and the mobile phase (moving).
Which chromatography technique is most commonly used?
High-Performance Liquid Chromatography (HPLC) is one of the most widely used techniques due to its high accuracy and efficiency.
Can chromatography be used for colorless compounds?
Yes, modern chromatography techniques are widely used for separating and analyzing colorless compounds.
What is the difference between stationary and mobile phase?
The stationary phase remains fixed and retains components, while the mobile phase moves and carries components through the system.

The Science Editorial Team creates clear, accurate, and student-friendly explanations of scientific concepts across biology, chemistry, physics, taxonomy, anatomy, and related life sciences. Content is written for educational and informational purposes using standard academic references and reliable sources. Read more about the → Science Editorial Team