Quick Answer: Thin layer chromatography (TLC) is widely used in chemistry to separate and identify non-volatile compounds.
It is applied in pharmaceuticals, food testing, pesticide analysis, medical diagnosis, cosmetic analysis, and biological research because it is simple, fast, and cost-effective.
What This Article Covers
- Basic principle of thin layer chromatography
- Applications in pharmaceuticals and medicine
- Role in food and pesticide analysis
- Use in biological and chiral studies
- Why TLC remains important despite advanced techniques
Core Concept: What Is Thin Layer Chromatography?
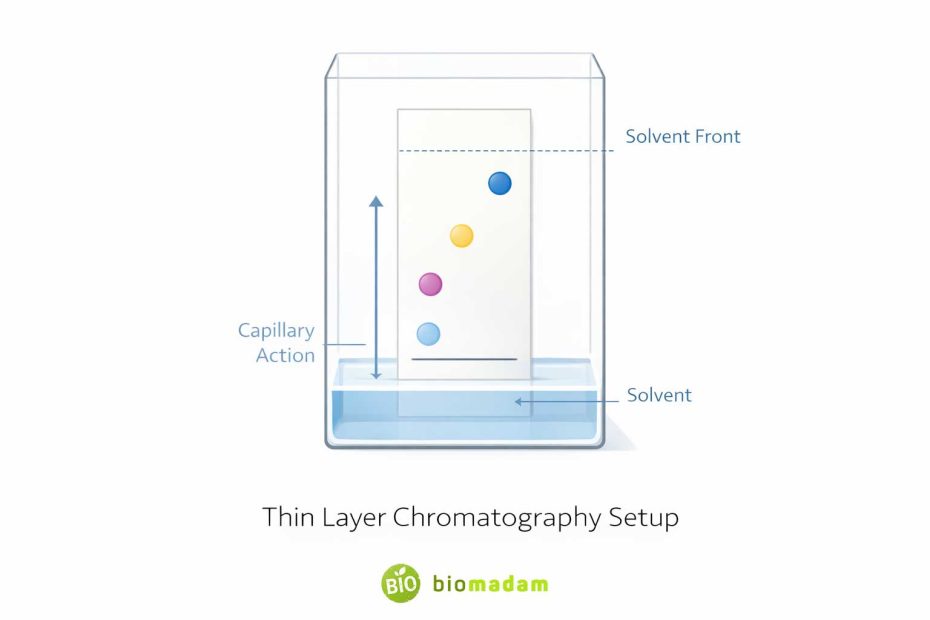
Thin layer chromatography (TLC) is a chromatographic technique used to separate components of a mixture.
The general separation mechanism is explained in Principle of Chromatography.
It consists of:
- Stationary phase: A thin layer of silica gel or alumina coated on a plate
- Mobile phase: A solvent or solvent mixture
When the plate is placed in the solvent, the liquid rises by capillary action. Different compounds move at different speeds depending on:
- Polarity
- Adsorption strength
- Solubility in the solvent
This difference allows separation and identification.
Understanding Rf Value in TLC
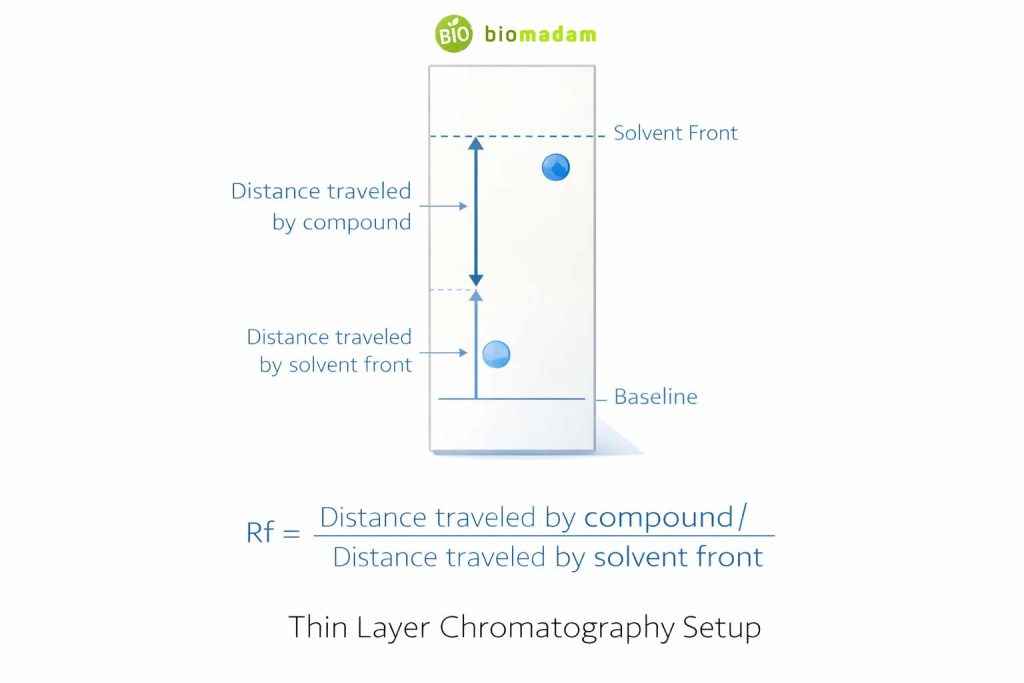
The retention factor (Rf value) is calculated as:
Rf = Distance traveled by compound / Distance traveled by solvent front
Rf values help identify compounds by comparing unknown samples with known standards under identical conditions.
Rf typically ranges between 0 and 1 and depends on solvent polarity and stationary phase.
Why Thin Layer Chromatography Is Important
TLC is widely used because it is:
- Rapid
- Inexpensive
- Easy to perform
- Suitable for small sample sizes
- Effective for preliminary screening
It is often used before advanced techniques such as HPLC or GC.
Because of its versatility, the practical uses of TLC extend across research laboratories, industrial quality control, and academic teaching environments.
Major Applications of Thin Layer Chromatography
Identification of Active Ingredients in Herbal and Traditional Medicines
Many traditional medicines are derived from plant extracts.
TLC helps:
- Separate plant metabolites
- Identify active compounds
- Compare unknown samples with known standards
By comparing Rf values (retention factor), researchers can confirm the presence of specific compounds in plant preparations.
Determination of Biological Activities
Antioxidant Activity
TLC is commonly used with DPPH (2,2-diphenyl-1-picrylhydrazyl) reagent.
Antioxidant compounds appear as yellow spots against a purple background, indicating radical scavenging activity.
Antimicrobial and Antifungal Activity
Separated compounds on a TLC plate can be tested against microorganisms.
Clear zones on the plate indicate inhibition of microbial growth.
This helps identify bioactive substances in plant extracts or synthetic compounds.
Chiral Analysis
In pharmaceutical research, distinguishing between enantiomers is critical.
Special TLC plates impregnated with chiral agents help separate optical isomers.
This is important because:
- One enantiomer may be therapeutically active
- The other may be less effective or inactive
Applications in Medical Diagnosis
TLC assists in detecting abnormal metabolites.
For example:
In porphyria, porphyrins are excreted in urine. TLC can separate and identify these compounds, aiding diagnosis.
Fluorescent detection under UV light enhances visibility.
Pharmaceutical Testing
Drug Stability Testing
TLC compares fresh and stored samples.
Changes in spot position or intensity indicate:
- Degradation
- Impurity formation
- Stability issues
For example, pharmaceutical manufacturers often use TLC as a quick quality control step before conducting more detailed HPLC analysis.
Drug Detection in Body Fluids
TLC is used in forensic and clinical analysis of:
- Blood
- Urine
- Plasma
It helps detect the presence of drugs and metabolites.
Purity Testing
TLC identifies impurities in:
- Antibiotics
- Steroids
- Analgesics
- Antihistamines
Additional spots indicate contamination.
Applications in the Pesticide Industry
TLC is widely used to:
- Detect pesticide residues
- Study pesticide degradation
- Analyze radiolabeled pesticide metabolism
- Monitor environmental contamination
It is especially useful for screening large numbers of samples quickly.
Applications in the Food Industry
TLC helps in:
- Detecting pesticide residues in food
- Identifying flavor compounds such as vanillin
- Detecting organic acids (benzoic acid, sorbic acid)
- Analyzing food additives and sweeteners
- Identifying mycotoxins such as aflatoxins
Because it is rapid and economical, TLC is often used in food quality control laboratories.
Detecting contaminants early helps prevent foodborne risks and ensures compliance with food safety regulations.
Applications in the Cosmetics Industry
TLC is used to:
- Analyze dyes and pigments
- Detect preservatives
- Identify active ingredients
- Monitor contamination
It supports quality assurance in cosmetic manufacturing.
Comparison: TLC vs Other Chromatographic Techniques
| Feature | Thin Layer Chromatography | HPLC | Gas Chromatography |
| Cost | Low | High | Moderate |
| Speed | Fast | Moderate | Fast |
| Sensitivity | Moderate | High | High |
| Quantitative Accuracy | Limited | High | High |
| Sample Volume Required | Very small | Moderate | Small |
| Instrument Requirement | Minimal | Advanced equipment | Advanced equipment |
TLC is ideal for screening, while HPLC and GC provide higher precision and sensitivity.
When Thin Layer Chromatography Is Preferred
Thin layer chromatography is often preferred when:
- Rapid screening is required
- Budget constraints limit advanced instrumentation
- Small sample quantities are analyzed
- Visual comparison of compounds is sufficient
It is frequently used as a preliminary analytical method before more advanced techniques.
Advantages of TLC in Practical Laboratories
- Simple procedure
- Low operational cost
- Visual interpretation
- Requires minimal equipment
- Suitable for educational demonstrations
These benefits make TLC widely used in academic institutions.
A detailed evaluation is available in Advantages and Disadvantages of Thin Layer Chromatography.
Limitations to Remember
Although versatile, TLC:
- Has lower sensitivity than HPLC
- Is less suitable for precise quantification
- May require visualization techniques (UV, staining reagents)
- Has limited automation
Understanding these limits helps choose the correct analytical method.
One-Line Summary
Thin layer chromatography is a simple and cost-effective analytical technique used to separate and identify compounds across pharmaceutical, food, pesticide, cosmetic, and research applications.
FAQs
1. What is thin layer chromatography mainly used for?
It is mainly used for separating and identifying non-volatile compounds.
2. Is TLC qualitative or quantitative?
TLC is primarily qualitative, though semi-quantitative analysis is possible.
3. Why is TLC preferred in teaching laboratories?
TLC is preferred because it is simple, visual, and inexpensive.
4. Can TLC detect pesticide residues?
Yes, it is widely used for pesticide residue screening.
5. What stationary phase is commonly used in TLC?
Silica gel is the most commonly used stationary phase.
6. What are the advantages of thin layer chromatography?
TLC is inexpensive, simple, rapid, and requires minimal instrumentation compared to advanced chromatographic techniques.
The Bottom Line
Thin layer chromatography remains an essential analytical tool due to its simplicity, affordability, and wide applicability. While advanced chromatographic methods offer greater sensitivity and automation, TLC continues to serve as a reliable first-line technique in laboratories worldwide.

The Science Editorial Team creates clear, accurate, and student-friendly explanations of scientific concepts across biology, chemistry, physics, taxonomy, anatomy, and related life sciences. Content is written for educational and informational purposes using standard academic references and reliable sources. Read more about the → Science Editorial Team