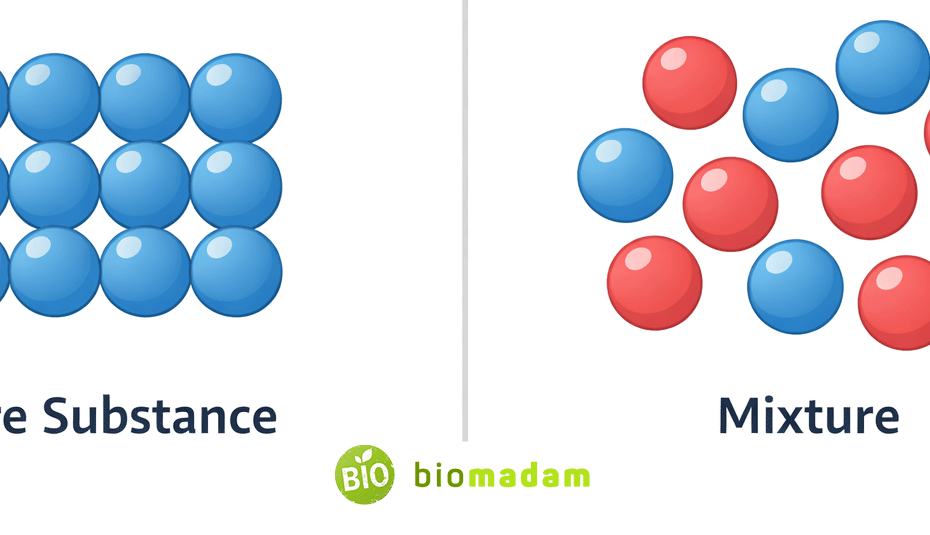
Quick Answer: A pure substance has a fixed chemical composition and uniform properties throughout, while a mixture consists of two or more substances physically combined in variable proportions. Pure substances cannot be separated into simpler substances by physical methods, though phase changes (melting/boiling) are physical processes, but mixtures can.
Comparison Between Pure Substance and Mixture
| Feature | Pure Substance | Mixture |
| Definition | Matter with fixed composition and definite properties | Physical combination of two or more substances |
| Composition | Fixed ratio of atoms | Variable composition |
| Types | Elements, Compounds | Homogeneous, Heterogeneous |
| Separation Method | Not separable by physical methods; may require chemical reactions | Physical methods (filtration, distillation, evaporation) |
| Melting/Boiling Point | Sharp and definite | Usually occurs over a range |
| Uniformity | Always uniform | May be uniform or non-uniform |
| Examples | Gold (Au), Water (H₂O), Sodium chloride (NaCl) | Air, Saltwater, Oil and water |
What is a Pure Substance?
A pure substance is a form of matter that has a constant chemical composition and characteristic properties. In chemistry, classification of matter into pure substances and mixtures is a foundational concept used to understand material behavior and reactions. It consists of only one type of particle — either atoms or molecules — arranged in a definite structure.
Understanding the structural distinction between particles is easier when reviewing the Difference between Molecules and Compounds.
Pure substances:
- Have fixed composition
- Exhibit uniform physical and chemical properties
- Cannot be separated into simpler substances by physical processes
- Have definite melting and boiling points
Pure substances are classified into two categories:
1. Elements
An element contains only one type of atom. It cannot be broken down into simpler substances by chemical means.
Examples:
- Hydrogen (H)
- Mercury (Hg)
- Lead (Pb)
- Oxygen (O₂)
2. Compounds
A compound consists of two or more different elements chemically bonded together in a fixed ratio.
Examples:
- Water (H₂O)
- Sodium chloride (NaCl)
- Carbon dioxide (CO₂)
Unlike mixtures, compounds form new substances with properties different from their constituent elements.
Important Clarification: All compounds are pure substances, but not all pure substances are compounds. Elements are also pure substances because they contain only one type of atom.
Why Pure Substances Have Fixed Properties
Pure substances contain identical particles arranged in a consistent structure. Because every particle is the same, their physical properties such as melting point, boiling point, and density remain constant throughout the sample. This uniformity is what distinguishes them from mixtures, where different particles disrupt consistency.
What is a Mixture?
A mixture is formed when two or more substances are physically combined without chemical bonding. Each component retains its original properties.
Mixtures:
- Have variable composition
- Can be separated by physical methods
- May be uniform or non-uniform
- Usually show melting/boiling ranges instead of fixed points
Examples:
- Air (mixture of gases)
- Saltwater
- Soil
- Salad
Types of Mixtures

1. Homogeneous Mixture
A homogeneous mixture has uniform composition throughout.
- Also called a solution
- Components are evenly distributed
- Cannot be visually distinguished
Example: Salt dissolved in water.
2. Heterogeneous Mixture
A heterogeneous mixture has non-uniform composition.
- Components are visibly distinct
- Particles may settle over time
Examples:
- Oil and water
- Sand in water
- Suspensions
For a detailed comparison, see the Difference Between Homogeneous & Heterogeneous Mixtures.
Core Concept: Fixed vs Variable Composition
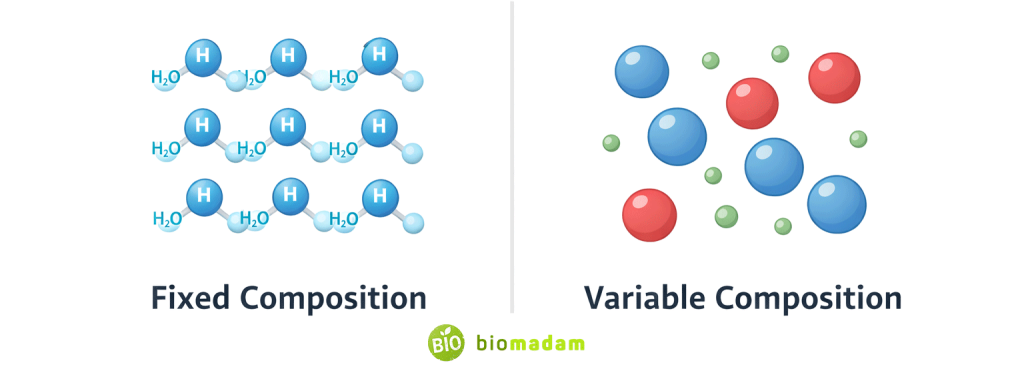
The most fundamental difference is:
- Pure substances have fixed atomic ratios.
- Mixtures have variable proportions.
For example: Water (H₂O) always contains hydrogen and oxygen in a 2:1 ratio.
This principle is explained by the Law of Definite Proportions, which states that a chemical compound always contains the same elements in the same fixed ratio by mass. This is why pure substances maintain constant composition regardless of sample size.
Saltwater may contain 1%, 5%, or 10% salt — the ratio is not fixed.
When to Use the Term Pure Substance vs Mixture?
Use pure substance when referring to:
- A single chemical compound
- An element
- Materials with constant properties
Use mixture when:
- Components can be physically separated
- Composition varies
- Substances retain individual properties
How to Identify Whether a Substance Is Pure or a Mixture
Ask these questions:
- Can it be separated using physical methods?
- Does it have a sharp melting or boiling point?
- Is the composition fixed?
- Are the particles chemically bonded?
If the answer supports fixed composition and chemical bonding, it is a pure substance. If it supports variability and physical separation, it is a mixture.
Separation Methods (Concept Depth)

Mixtures can be separated using:
- Filtration
- Distillation
- Evaporation
- Decantation
- Chromatography
Advanced separation techniques such as chromatography are used to separate components of complex mixtures based on differences in physical or chemical properties.
Understanding the Difference Between Physical and Chemical Properties further clarifies why mixtures can be separated without altering chemical identity.
Pure substances require chemical reactions to break into simpler substances.
Examples:
- Water can be decomposed into hydrogen and oxygen only through electrolysis (a chemical process).
- A mixture of iron and sand can be separated using a magnet.
- Saltwater can be separated by evaporation or distillation.
- Oil and water can be separated using a separating funnel.
These processes do not alter chemical identity, which confirms the material is a mixture.
This distinction also aligns with the Difference Between Physical And Chemical Change, where physical processes do not modify chemical composition.
Common Confusion Alert
Many students confuse the Difference between Compounds and Mixture, especially when distinguishing chemical bonding from physical combination.
- A compound forms through chemical bonding and has new properties.
- A mixture does not involve chemical bonding and retains original properties.
Simple Analogy to Understand the Difference
A pure substance is like a box of identical blue Lego blocks — every piece is the same.
A mixture is like a box containing red, blue, and yellow Lego blocks randomly combined.
The uniformity of pieces represents fixed composition, while mixed colors represent variable composition.
Key Differences Summary
- Pure substances → fixed composition, definite properties
- Mixtures → variable composition, separable physically
- Pure substances → sharp melting/boiling point
- Mixtures → melting/boiling range
- Pure substances → chemical bonds present
- Mixtures → no chemical bonding
FAQs
Is air a pure substance or a mixture?
Air is a mixture of gases including nitrogen, oxygen, and carbon dioxide. Its composition can vary slightly depending on location and altitude.
Can mixtures be separated easily?
Most mixtures can be separated using physical methods like filtration or distillation. However, some mixtures such as azeotropes require special separation techniques.
Is saltwater a pure substance?
No. Saltwater is a homogeneous mixture because salt and water are physically combined and can be separated by evaporation.
Do pure substances always have fixed melting points?
Yes. Pure substances exhibit sharp and definite melting and boiling points under standard conditions.
The Bottom Line
Understanding the difference between pure substances and mixtures is fundamental in chemistry because it forms the basis for classification of matter, chemical reactions, and laboratory analysis. The defining distinction lies in fixed chemical composition versus variable physical combination.

The Science Editorial Team creates clear, accurate, and student-friendly explanations of scientific concepts across biology, chemistry, physics, taxonomy, anatomy, and related life sciences. Content is written for educational and informational purposes using standard academic references and reliable sources. Read more about the → Science Editorial Team