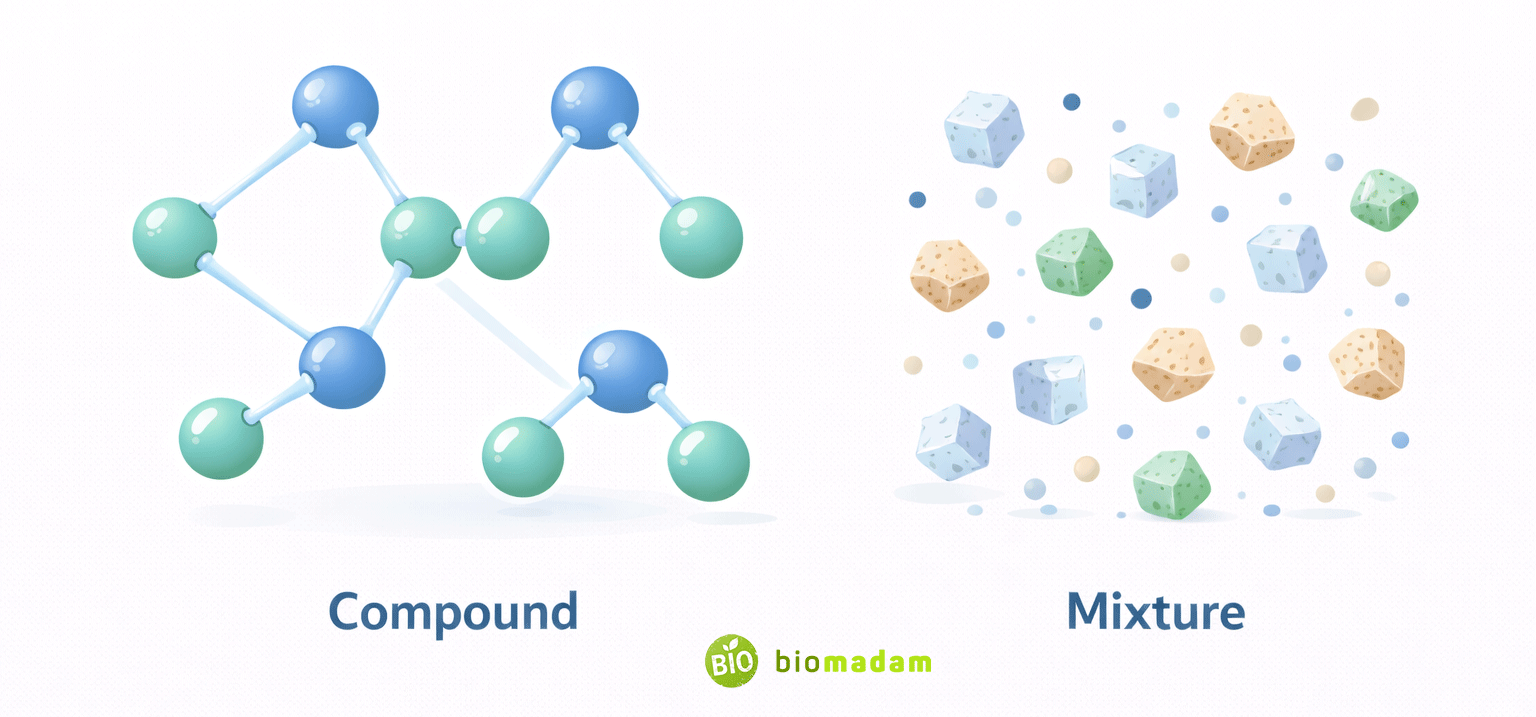
Quick Answer: A compound is a pure substance formed when two or more elements chemically bond together in a fixed ratio, creating a new substance with unique properties. A mixture is a physical combination of two or more substances in variable proportions, where each component retains its original properties.
This distinction is fundamental in chemistry because it determines how substances behave, react, and can be separated.
The main difference is that compounds involve chemical bonding and fixed composition, whereas mixtures involve physical combination and variable composition.
Comparison Between Compounds and Mixtures
| Feature | Compound | Mixture |
| Definition | Substance formed by chemical bonding of elements | Physical combination of two or more substances |
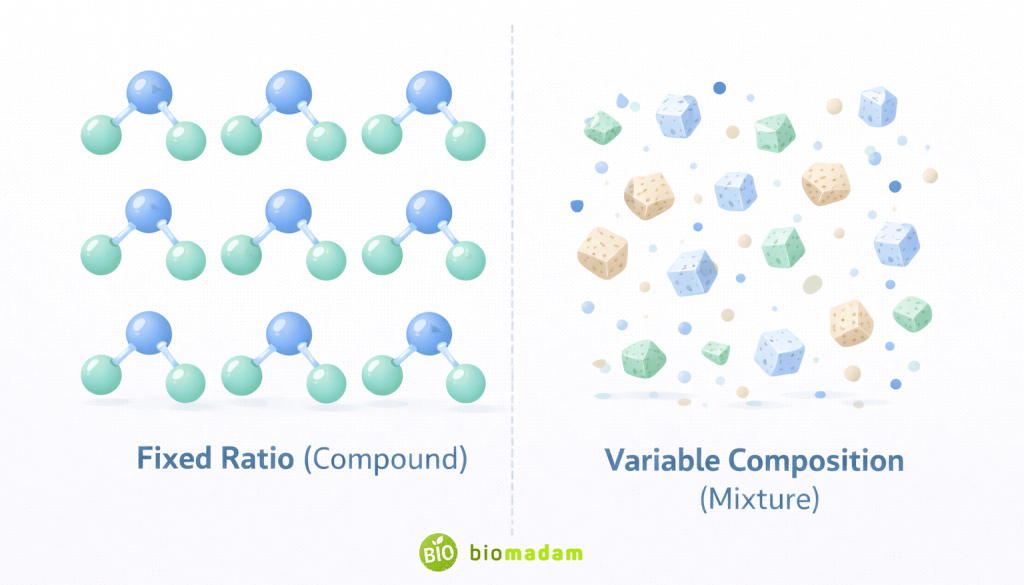
| Composition | Fixed ratio by mass | Variable proportions |
| Type of Bonding | Chemical bonds present | No chemical bonding between components |
| Separation Method | Chemical reactions required | Physical methods sufficient |
| Properties | New properties different from elements | Components retain original properties |
| Melting/Boiling Point | Definite and sharp | Usually occurs over a range |
| Representation | Chemical formula (e.g., H₂O) | No fixed chemical formula |
| Nature | Pure substance | May be homogeneous or heterogeneous |
| Examples | Water (H₂O), NaCl, CO₂ | Air, saltwater, oil and water |
To better understand how pure substances differ from mixtures at a broader level, see the Difference Between Pure Substance and Mixture.
Compound vs Mixture in One Line
- Compound → chemically bonded, fixed ratio, new properties
- Mixture → physically combined, variable ratio, original properties retained
Direct Difference Between Compounds and Mixtures
A compound forms when elements chemically react and bond in a fixed ratio, producing a new substance with different properties. A mixture forms when substances are physically combined without chemical bonding, so each substance maintains its original identity.
What is a Compound?
A compound is a pure substance composed of two or more different elements chemically bonded together in a fixed proportion by mass. Compounds are formed through chemical reactions in which energy is absorbed or released due to bond formation or bond breaking. This energy change reflects the stability of the new substance formed.
Key characteristics:
- Fixed composition
- Chemical bonds present (ionic or covalent)
For a deeper explanation of bond types, refer to the Difference between Ionic, Covalent and Metallic Bonds.
- Definite melting and boiling points
- Represented by a chemical formula
- Cannot be separated by physical means
Example: Water (H₂O) always contains hydrogen and oxygen in a 2:1 ratio.
This fixed ratio follows the Law of Definite Proportions, which states that a compound always contains the same elements in the same proportion by mass.
Compound formation also follows the Law of Conservation of Mass, which states that matter is neither created nor destroyed during a chemical reaction, but rearranged into new chemical identities.
Types of Compounds
Ionic Compounds
Formed by transfer of electrons between metals and nonmetals.
Example:
- Sodium chloride (NaCl)
These often form crystal lattices.
Covalent Compounds
Formed when atoms share electrons.
Examples:
- Carbon dioxide (CO₂)
- Methane (CH₄)
Also known as molecular compounds; see the Difference between Molecules and Compound for structural clarification.
Organic and Inorganic Compounds
- Organic compounds contain carbon bonded to hydrogen (e.g., glucose, proteins).
- Inorganic compounds generally lack carbon–hydrogen bonds (e.g., NaCl, CO₂, NH₃).
What is a Mixture?
A mixture is a physical combination of two or more substances where no chemical reaction occurs and each component retains its properties.
Key characteristics:
- Variable composition
- No chemical bonding between components
- Can be separated by physical methods
- No chemical formula representation
Examples:
- Air (mixture of gases)
- Salt dissolved in water
- Sand and water
Types of Mixtures
Homogeneous Mixtures
- Uniform composition
- Single phase
- Also called solutions
Example: Saltwater (solute: salt; solvent: water)
A structured comparison is available in the Difference Between Homogeneous and Heterogeneous Mixtures.
Heterogeneous Mixtures
- Non-uniform composition
- Two or more phases
- Components may be visibly distinguishable
Examples:
Oil and water
Sand in water
Core Concept: Chemical Bonding vs Physical Mixing
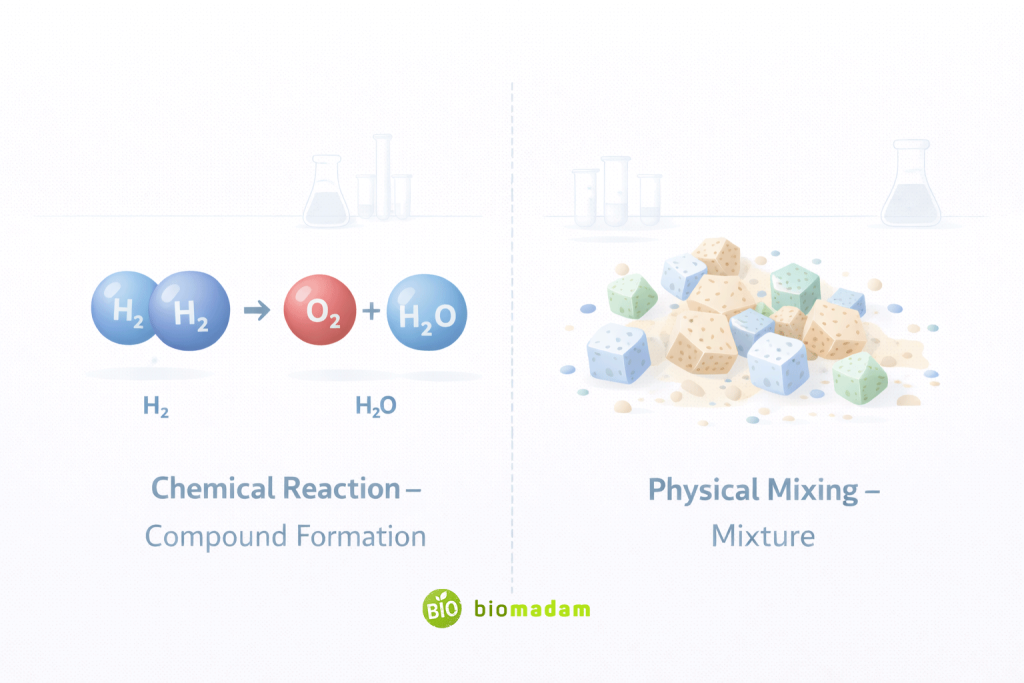
The fundamental distinction lies in bonding.
- In compounds, atoms are chemically bonded, forming new substances.
This bonding results in a new chemical identity and often involves stoichiometric ratios that define the precise composition of the compound.
- In mixtures, substances are only physically combined.
For example:
- Hydrogen and oxygen chemically react to form water (compound).
- Salt and sand mixed together remain salt and sand (mixture).
Separation Differences
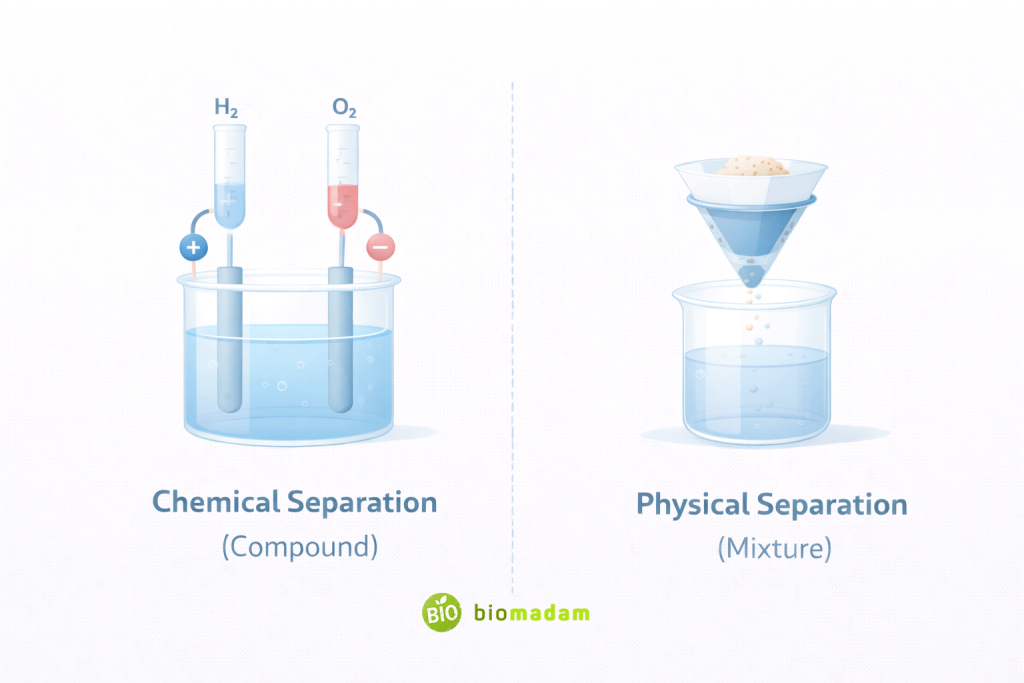
Compounds:
- Require chemical reactions (e.g., electrolysis of water)
This distinction reflects the Difference Between Physical And Chemical Change, where chemical changes alter substance identity while physical changes do not.
Mixtures:
- Filtration
- Distillation
- Evaporation
- Magnetic separation
This difference determines laboratory separation techniques.
How to Identify Compound or Mixture
Ask:
- Is there a chemical formula?
- Is the composition fixed?
- Are new properties formed?
- Can it be separated physically?
If new properties and fixed ratio exist → compound.
If components retain properties and can be separated physically → mixture.
Is One More Stable Than the Other?
Compounds generally have stable chemical structures due to bonding between atoms. Mixtures do not involve chemical bonding between components, so their stability depends on the individual substances present rather than a new chemical structure.
Common Confusion Alert
- Milk is a mixture (colloid), not a compound.
- Saltwater is a mixture, not a compound.
- Water (H₂O) is a compound, not a mixture.
The presence of more than one element does not automatically make something a mixture — chemical bonding is the deciding factor.
Key Differences Summary
- Compounds → chemical bonds, fixed ratio, pure substances
- Mixtures → physical combination, variable ratio
- Compounds → new properties formed
- Mixtures → original properties retained
- Compounds → separated chemically
- Mixtures → separated physically
Real-Life Examples Explained
- Steel is a homogeneous mixture (alloy of iron and carbon).
- Milk is a colloid and therefore a heterogeneous mixture.
- Carbon dioxide (CO₂) is a compound because carbon and oxygen are chemically bonded.
- Brass is a mixture of copper and zinc.
FAQs
Is air a compound or a mixture?
Air is a mixture because it consists of different gases physically combined without chemical bonding.
Is saltwater a compound?
No. Saltwater is a homogeneous mixture because salt and water can be separated physically by evaporation.
Can mixtures form new substances?
No. Mixtures do not form new chemical substances; their components retain original identities.
Do compounds always have fixed composition?
Yes. Compounds follow the Law of Definite Proportions and have fixed mass ratios.
Is milk a compound or mixture?
Milk is a mixture because it consists of water, fats, and proteins physically dispersed as a colloid.
Is blood a compound or mixture?
Blood is a heterogeneous mixture composed of plasma, red blood cells, white blood cells, and platelets.
Is steel a compound or mixture?
Steel is a homogeneous mixture (alloy) of iron and carbon, not a compound.
Can a compound become a mixture?
Yes. A compound can become part of a mixture if it is physically combined with other substances without undergoing a chemical reaction.
The Bottom Line
Compounds and mixtures differ fundamentally in chemical bonding and composition. Compounds are pure substances formed by chemical reactions in fixed ratios, while mixtures are physical combinations of substances in variable proportions. Understanding this distinction is essential for classification of matter, chemical reactions, and separation processes.

The Science Editorial Team creates clear, accurate, and student-friendly explanations of scientific concepts across biology, chemistry, physics, taxonomy, anatomy, and related life sciences. Content is written for educational and informational purposes using standard academic references and reliable sources. Read more about the → Science Editorial Team