Carbon is an essential part of many compounds and mixtures around us. Two of the most common carbon compounds are carbon dioxide and carbon monoxide. You must have heard about carbon dioxide as one of the gases in our atmosphere. It is released by animals during breathing and utilized by plants for photosynthesis. On the other hand, carbon monoxide is present in gas heaters, boilers, fossil fuels, etc. While the structural difference between carbon dioxide and carbon monoxide is only one oxygen atom, it has a huge impact on their properties. Let’s tell you further about both of them.
Comparison Table
| Characteristics | Carbon Dioxide | Carbon Monoxide |
| Composition | 1 carbon + 2 oxygen | 1 carbon + 1 oxygen |
| Chemical Formula | CO2 | CO |
| Bonds | Covalent | Covalent + coordinate |
| Molecular Mass | 44 g/mol | 28.1 g/mol |
| Origin | Natural | Synthetic |
| Source | Fossil fuels, organic matter | Motor vehicles, heaters |
| Flammability | Non-flammable | Flammable |
| Application | Food & beverage industries | Reducing agent |
| Safe Level | 5000 ppm | 50 ppm |
| Toxic Effects | Respiratory system | Nervous system |
What is Carbon Dioxide?
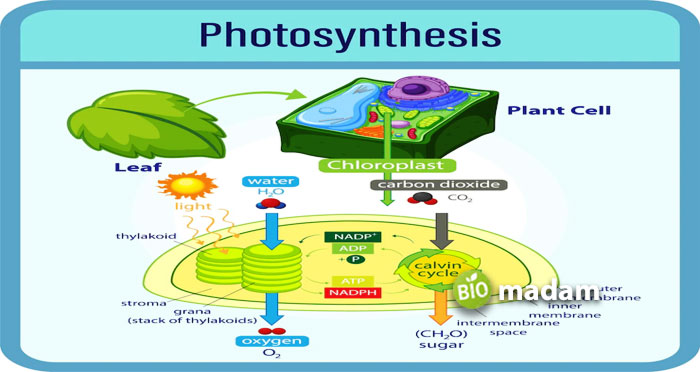
Carbon dioxide is an essential biological compound formed by carbon and oxygen. Each carbon dioxide molecule contains one atom of carbon and two oxygen atoms. It is an odorless, tasteless and colorless gas at room temperature. Carbon dioxide is non-flammable and tests faintly acidic. The chemical formula for carbon dioxide is CO2. The carbon atom forms a linear chain in each molecule by double bonds to the oxygen atoms on each side.
Carbon dioxide is widely found in the atmosphere. The concentration is enough for the plants to carry out photosynthesis and not harm humans and animals. It comes from the decomposition of organic matter and the burning of fossil fuels. Food freezing and beverage industries use carbon dioxide. Other applications are in the medical and agricultural industries.
Carbon Dioxide Poisoning
The safe carbon dioxide levels in the atmosphere are way higher than carbon monoxide. Still, an unrealistically high concentration of CO2 can cause poisoning.
- ASHRAE recommends maintaining a 1000 ppm limit for schools and office buildings for health safety measures.
- Workplace carbon dioxide levels should not be any more than 5,000 ppm for an 8-hour workday.
- 10,000 ppm (1%) can lead to drowsiness and fainting in closed areas.
- Mild poisoning occurs at 30,000 ppm (3%) with headaches and dizziness.
- Carbon dioxide can cause death at a 4% concentration (40,000 ppm).
What is Carbon Monoxide?
It is pretty easy to confuse carbon monoxide with carbon dioxide as both are odorless and colorless. However, both have vastly different chemical properties, applications, and toxicity. Carbon Monoxide is also composed of carbon and oxygen. As opposed to carbon dioxide, carbon monoxide contains one atom of carbon and oxygen each. Carbon monoxide is not present in the atmosphere naturally. Instead, it is a synthetic gas used in many gadgets and machinery. The carbon monoxide chemical formula is CO. It contains a triple bond between the two atoms. Two bonds are covalent, whereas one is a coordinate bond.
Motor vehicles and heaters running on organic matter produce carbon monoxide. It forms ozone at ground level. Yet, carbon monoxide has a short lifespan in the atmosphere. High concentrations of carbon monoxide above 35 ppm can be harmful to life. It might surprise you that carbon monoxide poisoning is the most common fatal poisoning around the globe. It is also named the “silent killer” because you cannot detect increasing CO levels without gas detectors.
Carbon Monoxide Poisoning
Carbon monoxide is a highly poisonous gas and must not be inhaled in a high concentration. No more than 9-50 ppm carbon monoxide must be present for an 8-hour workday. CO poisoning can cause headaches, vomiting, and dizziness leading to death.
- Carbon Monoxide concentrations up to 100 ppm cause mild CO poisoning.
- Between 200-400 ppm, of carbon monoxide, will lead to unconsciousness and death within a few hours.
- CO concentrations of more than 800ppm can prove fatal within minutes.

Similarities Between Carbon Dioxide and Carbon Monoxide
- Carbon dioxide and carbon monoxide contain carbon and oxygen.
- Both of them are gases.
- They are colorless, tasteless, and odorless.
- These gases are potentially toxic in high concentrations.
Differences Between Carbon Dioxide And Carbon Monoxide
Definition
Carbon Dioxide
Carbon dioxide is a gas present in the atmosphere in a moderate concentration.
Carbon Monoxide
Alternatively, carbon monoxide is a synthetic gas known as the silent killer.
Composition
Carbon Dioxide
Carbon dioxide is made of one carbon atom and two oxygen atoms.
Carbon Monoxide
On the other hand, carbon monoxide contains one carbon and one oxygen atom.
Chemical Formula
Carbon Dioxide
The chemical formula for carbon dioxide is CO2.
Carbon Monoxide
Whereas the chemical formula for carbon monoxide is CO.
Bonds
Carbon Dioxide
Carbon dioxide bonds to the two oxygen atoms through a covalent (double) bond.
Carbon Monoxide
Yet, carbon monoxide has triple bonds. It contains covalent bonds and coordinates covalent bonds.
Molecular Mass
Carbon Dioxide
The molecular mass of carbon dioxide is 44 g/mol.
Carbon Monoxide
On the contrary, carbon monoxide’s molecular mass is 28.1 g/mol.
Origin
Carbon Dioxide
The origin is a critical difference between carbon dioxide and carbon monoxide. Carbon dioxide is a natural gas found in the earth’s atmosphere.
Carbon Monoxide
Conversely, carbon monoxide is a synthetic gas produced by the combustion of the material.
Source
Carbon Dioxide
Carbon Dioxide emits from burning fossil fuels and the decomposition of organic matter.
Carbon Monoxide
Unlike, carbon monoxide comes from motor vehicles, cookers, and heaters burning on organic material.
Flammability
Carbon Dioxide
Carbon dioxide is a non-flammable gas.
Carbon Monoxide
But, carbon monoxide is a flammable gas.
Applications
Carbon Dioxide
Carbon dioxide is used in the agricultural, medical, and beverage industries.
Carbon Monoxide
Carbon monoxide is used as a reducing agent in different industries.
Safe Levels
Carbon Dioxide
Carbon dioxide is safe at up to 5000 ppm for an 8-hour workday.
Carbon Monoxide
Nevertheless, carbon monoxide can be toxic at above 50 ppm for an 8-hour workday.
Toxic Effects
Carbon Dioxide
Carbon dioxide can affect the respiratory system when inhaled in high concentrations.
Carbon Monoxide
As opposed to CO2, carbon monoxide affects the central nervous system at a toxic level.
The Bottom Line
Carbon dioxide and carbon monoxide are carbon compounds. They both contain oxygen. CO and CO2 are colorless, odorless, and tasteless gases, but their chemical formula and properties differ. Carbon dioxide is present in the earth’s atmosphere, while carbon monoxide comes from the combustion of heaters, cookers, etc. The toxic effects of both gases constitute a significant difference between carbon dioxide and carbon monoxide. Carbon dioxide is a harmful gas, whereas carbon monoxide is fatal. A slight increase in the concentration of carbon monoxide can prove life-threatening.
FAQs
Is carbon dioxide dangerous?
Prolonged exposure to carbon dioxide in large quantities can cause several health issues. They include restlessness, headache, dizziness, sweating, tiredness, increased heart rate, and blood pressure, a tingling needle-like feeling, and convulsions.
What are the symptoms of carbon monoxide poisoning?
The symptoms of carbon monoxide poisoning are “flu-like.” You may experience weakness, headache, and dizziness. Chest pain, vomiting, and confusion depict a worsening condition.
Is carbon monoxide or carbon dioxide worse for the environment?
Carbon dioxide is harmful but not as dangerous as carbon monoxide. CO is a synthetic gas. Raised levels of carbon monoxide can cause the death of an organism within minutes.
Do cars emit carbon monoxide or carbon dioxide?
Cars emit carbon monoxide when burning fuel. Do not stay in an enclosed car for a long time, especially when using the heater. Slide the windows after a few minutes or hours to avoid carbon monoxide accumulation in the vehicle.
Do humans exhale carbon monoxide or carbon dioxide?
Humans inhale oxygen and exhale carbon dioxide during external respiration, a type of respiration. Plants use it in photosynthesis and release oxygen. Over-inhalation of carbon dioxide is harmful to health.

The Biomadam Content Team develops and manages educational and informational articles published on Biomadam. Content is prepared using standard reference materials and follows internal editorial guidelines to ensure clarity and consistency.
