Quick Answer: The main difference between conductors and insulators is their ability to allow electric current to flow.
Conductors have free electrons and overlapping energy bands, resulting in low resistance and high conductivity.
Insulators have tightly bound electrons and a large forbidden energy gap, resulting in very high resistance and extremely low conductivity under normal conditions.
Comparison Between Conductors and Insulators
| Feature | Conductors | Insulators |
| Definition | Materials that allow electric current to flow easily | Materials that resist the flow of electric current |
| Charge Carriers | Many free electrons | Almost no free charge carriers |
| Electron Movement | Free movement of electrons | Electrons tightly bound to atoms |
| Electrical Resistance | Low | Very High |
| Conductivity | High | Extremely low |
| Energy Band Gap | No band gap or overlapping bands | Large forbidden energy gap |
| Temperature Effect | Resistance increases with temperature (positive TCR) | Resistance decreases with temperature (negative TCR, generally) |
| Electric Field Inside | Zero at electrostatic equilibrium | Can exist inside material |
| Examples | Copper, silver, aluminum, graphite | Glass, rubber, plastic, dry wood, air |
| Uses | Electrical wiring, circuits, power transmission | Wire coating, insulation, safety barriers |
| Typical Resistivity | Very low (e.g., copper ≈ 1.7 × 10⁻⁸ Ω·m) | Extremely high (e.g., glass ≈ 10¹⁰–10¹⁴ Ω·m) |
| Energy Band Structure | Valence and conduction bands overlap | Large energy band gap |
| Electric Field Inside (Electrostatic Equilibrium) | Zero | May exist |
| Thermal Conductivity | Generally high (in metals) | Generally low |
| Breakdown Behavior | Conduct easily under normal voltage | Conduct only at high breakdown voltage |
What Are Conductors?
Conductors are materials that allow electric current to flow easily due to the presence of free electrons or charge carriers.
In metals, outer-shell (valence) electrons are loosely bound and can move freely when a potential difference is applied. This movement of electrons creates electric current, a fundamental concept studied in physics, particularly in electricity and solid-state theory.
Band Theory Explanation
In conductors:
- The valence band and conduction band overlap
- There is no forbidden energy gap
- Electrons can move freely without needing extra energy
This makes metals excellent electrical conductors. The behavior of electrons in conductors is explained through band theory in solid-state physics, which describes how energy levels determine electrical properties.
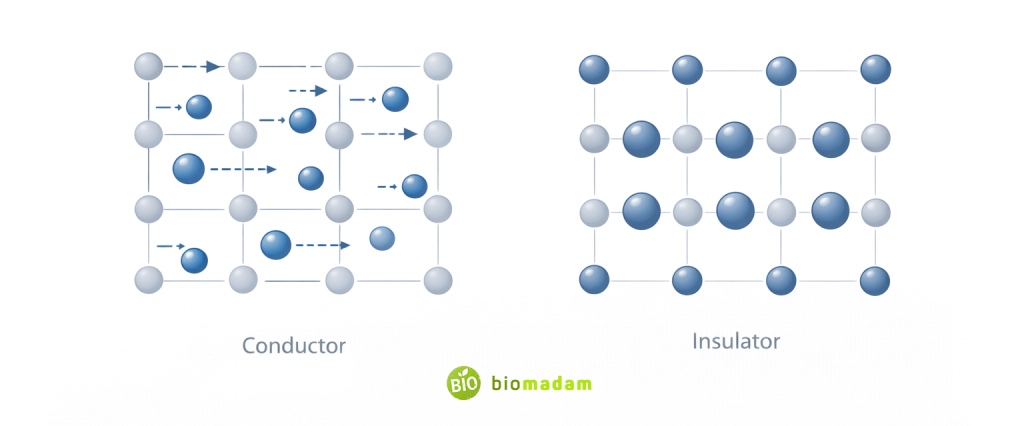
Properties of Conductors
- High electrical conductivity
- Low resistance
- Free electron movement
- Electric field inside is zero (at electrostatic equilibrium)
- Positive temperature coefficient of resistance
Temperature Behavior
As temperature increases, metal atoms vibrate more, causing more collisions with electrons.
This increases resistance.
Examples of Conductors
- Copper
- Silver
- Aluminum
- Iron
- Graphite (non-metal but conducts electricity)
What Are Insulators?
Insulators are materials that strongly resist the flow of electric current because they lack free charge carriers.
In these materials, electrons are tightly bound to atoms and cannot move freely under normal conditions.
Band Theory Explanation
In insulators:
- The valence band and conduction band are separated
- There is a large forbidden energy gap
- Electrons require significant energy to jump into the conduction band
Because of this large band gap, current cannot flow easily.
Properties of Insulators
- Very high resistance
- Very low conductivity
- Large band gap
- Electrons tightly bound
- Generally negative temperature coefficient
Breakdown Voltage
If voltage becomes extremely high, an insulator may suddenly conduct electricity.
This is called dielectric breakdown.
Electrical Resistivity Comparison
Electrical resistivity measures how strongly a material opposes electric current.
- Copper resistivity ≈ 1.7 × 10⁻⁸ Ω·m
- Aluminum resistivity ≈ 2.8 × 10⁻⁸ Ω·m
- Glass resistivity ≈ 10¹⁰–10¹⁴ Ω·m
This massive difference explains why metals conduct electricity efficiently while materials like glass and rubber do not.
Examples of Insulators
- Glass
- Rubber
- Plastic
- Dry wood
- Ceramic
- Air (under normal conditions)
Key Concept: Energy Band Gap Difference
The fundamental difference between conductors and insulators lies in their energy band structure.
- Conductors → Overlapping bands → Free electron movement
- Insulators → Large band gap → Restricted electron movement
This microscopic difference determines macroscopic electrical behavior. Similar microscopic particle behavior is discussed in Difference Between Diffusion and Effusion, where particle motion explains macroscopic gas properties.
Where Do Semiconductors Fit?
Between conductors and insulators lies a third category called semiconductors.
- They have a small energy band gap.
- Their conductivity is moderate.
- Conductivity increases significantly with temperature or doping.
- Silicon and germanium are common examples.
Semiconductors are essential in modern electronics, forming the basis of transistors, diodes, and microchips.
Real-World Applications
Applications of Conductors
- Electrical wiring
- Power transmission lines
- Electronic circuits
- Motors and generators
Applications of Insulators
- Wire coatings (PVC, rubber)
- Transformer insulation
- Electrical poles and supports
- Building thermal insulation
Why Electrical Wires Use Both Conductors and Insulators

Electrical wires are made of copper (conductor) to allow efficient current flow.
They are coated with plastic or rubber (insulators) to prevent electric shock and short circuits.
This combination ensures both efficiency and safety in electrical systems.
Common Misconceptions
- Insulators do contain electric charges, but they do not have free-moving charges.
- Water is not always a conductor — pure distilled water is a poor conductor; impurities make it conductive.
- Glass can conduct electricity at very high temperatures.
- No material is a perfect insulator; extreme voltage can cause breakdown.
- In conductors, excess electric charge resides on the surface at electrostatic equilibrium, not inside the material.
Similarities Between Conductors and Insulators
- Both are made of atoms and contain electrons
- Both respond to electric fields
- Both can conduct electricity under extreme conditions
- Both are essential in electrical engineering
FAQs
What is the main difference between conductors and insulators?
Conductors allow free movement of electrons, while insulators restrict electron movement due to a large energy band gap.
Is water a conductor or insulator?
Pure water is a poor conductor. However, tap water conducts electricity because it contains dissolved ions.
Why do metals conduct electricity?
Metals have overlapping valence and conduction bands, allowing free electrons to move easily.
Can an insulator become a conductor?
Yes. Under high voltage (breakdown voltage) or high temperature, insulators can conduct electricity.
What is an example of a good conductor?
Copper is one of the best conductors used in electrical wiring.
Are humans conductors or insulators?
The human body is a conductor because it contains water and dissolved ions that allow electric current to pass.
Why is rubber used as an insulator?
Rubber has a large energy band gap and high resistance, preventing free electron movement.
What is the difference between conductors, insulators, and semiconductors?
Conductors have overlapping energy bands, insulators have large band gaps, and semiconductors have small band gaps that allow controlled conductivity.
Scientific Comparison Summary
- Conductors → Free electrons → Low resistance
- Insulators → Large band gap → High resistance
- Conductors → Overlapping energy bands
- Insulators → Wide forbidden energy gap
- Conductors → Positive TCR
- Insulators → Generally negative TCR
Conclusion
Conductors and insulators differ primarily in their ability to allow electric charge flow. Conductors contain free electrons that move easily due to overlapping energy bands, resulting in high conductivity and low resistance. Insulators, on the other hand, have tightly bound electrons and a large forbidden energy gap, preventing current flow under normal conditions. Understanding this difference is essential in physics, electrical engineering, and modern electronics.

The Science Editorial Team creates clear, accurate, and student-friendly explanations of scientific concepts across biology, chemistry, physics, taxonomy, anatomy, and related life sciences. Content is written for educational and informational purposes using standard academic references and reliable sources. Read more about the → Science Editorial Team
