Quick Answer: No, ionic bonds do not share electrons.
Instead, electrons are transferred from one atom to another, forming oppositely charged ions that attract through electrostatic forces.
Electron sharing occurs only in covalent bonds.
What This Article Covers
- Whether ionic bonds share electrons
- How ionic bonds are formed
- How covalent bonds differ
- Ionic vs covalent comparison
- Real examples
- Why this difference matters
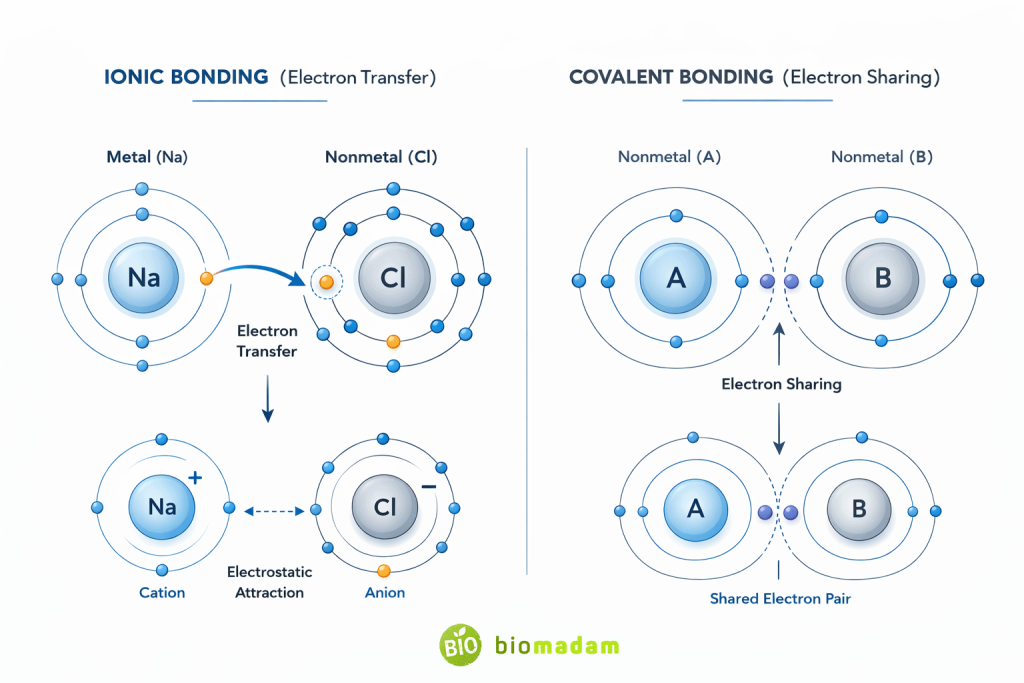
Core Concept: Electron Transfer vs Electron Sharing
Chemical bonds form when atoms interact to achieve a stable electron configuration.
An ionic bond is the electrostatic attraction between oppositely charged ions formed after electron transfer.
There are two main ways this happens:
- Electron transfer → Ionic bonding
- Electron sharing → Covalent bonding
A broader comparison is explained in Difference between Ionic, Covalent and Metallic Bonds.
The key difference is simple:
- Ionic bonds involve complete transfer of electrons.
- Covalent bonds involve sharing of electrons between atoms.
Understanding this distinction explains many physical and chemical properties of substances.
How Are Ionic Bonds Formed?
Ionic bonds form when:
- One atom loses one or more electrons.
- Another atom gains those electrons.
- Oppositely charged ions attract each other.
Atoms tend to achieve a stable configuration similar to noble gases (often following the octet rule, although there are exceptions).
- Elements in Groups 1 and 2 (metals) usually lose electrons → form cations (+)
- Elements in Groups 15–17 (nonmetals) usually gain electrons → form anions (−)
The electrostatic attraction between these opposite charges creates the ionic bond.
The formation of these charged species is explained in Difference Between Cations and Anions.
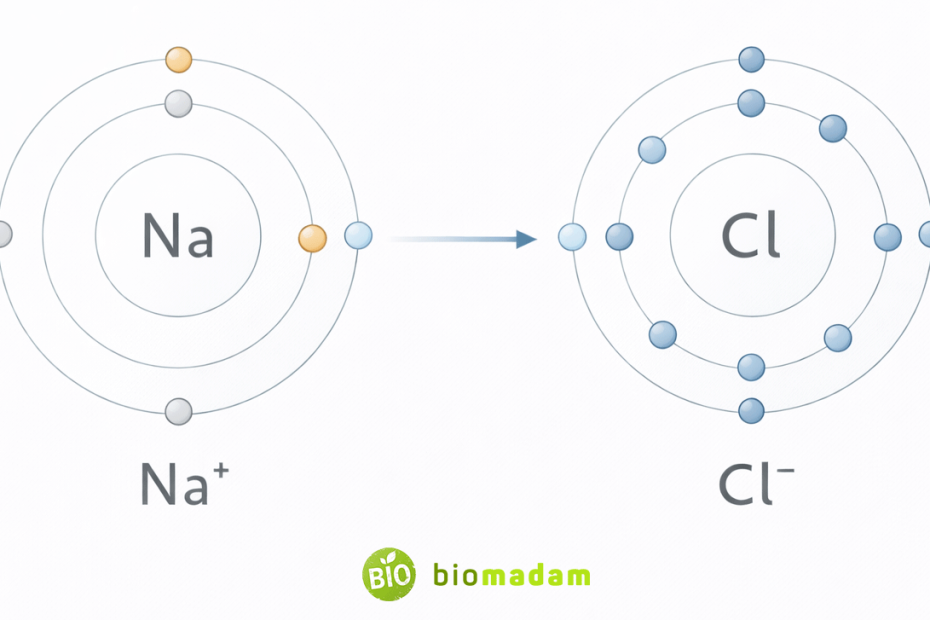
Example: Sodium and Chlorine
Sodium (Na):
- Has 1 valence electron
- Easily loses 1 electron → becomes Na⁺
The process of positive ion formation is detailed in How Are Cations Formed?
Chlorine (Cl):
- Has 7 valence electrons
- Gains 1 electron → becomes Cl⁻
Na transfers an electron to Cl.
This forms:
- Na⁺
- Cl⁻
These oppositely charged ions attract each other, forming sodium chloride (NaCl).
No electron sharing occurs — only transfer.
Why Ionic Bonds Do Not Share Electrons
In ionic bonding:
- Ionic bonding typically occurs when the electronegativity difference between atoms is large (generally greater than 1.7 on the Pauling scale).
- One atom attracts electrons much more strongly than the other.
- As a result, the electron is effectively transferred rather than shared.
After transfer:
- The resulting cations and anions attract each other through Coulombic (electrostatic) forces.
- The bond is maintained by charge attraction, not shared orbitals.
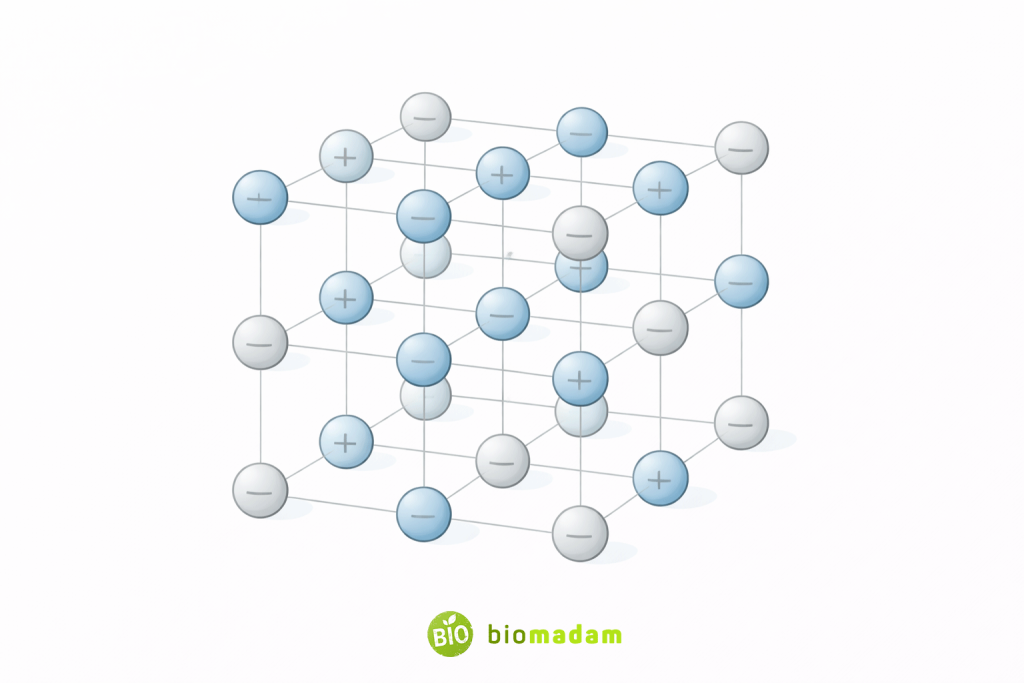
In solid ionic compounds, ions arrange in a repeating three-dimensional lattice structure rather than forming individual discrete molecules.
If ionic bonding involves electron transfer, what happens when atoms have similar attraction for electrons?
In that case, atoms tend to share electrons instead.
Do Covalent Bonds Share Electrons?
Yes.
Covalent bonds form when two atoms share electrons to complete their outer shells.
This typically occurs between nonmetals with similar electronegativity values.
Types of covalent bonds:
- Single bond → 1 shared pair
- Double bond → 2 shared pairs
- Triple bond → 3 shared pairs
Example: Methane (CH₄)
Carbon shares electrons with hydrogen atoms.
Here, electrons are shared — not transferred.
A related concept is discussed in Do Hydrogen Bonds Share Electrons?
Ionic vs Covalent Bonds (Clear Comparison)
| Feature | Ionic Bond | Covalent Bond |
| Electron Behavior | Electrons are transferred | Electrons are shared |
| Elements Involved | Usually metal + nonmetal | Usually nonmetal + nonmetal |
| Charge Formation | Forms ions (cations and anions) | No full charge formation |
| Melting Point | Often high due to strong electrostatic forces in ionic lattices | Usually lower (except network solids) |
| Example | NaCl | CH₄ |
Despite their differences, some similarities are discussed in Similarities between Ionic and Covalent Bonds.
Why This Difference Matters
The difference between transfer and sharing explains:
- Melting point → Ionic compounds often have high melting points.
- Electrical conductivity → Ionic compounds conduct electricity when molten or dissolved in water.
- Solubility → Ionic compounds dissolve well in polar solvents like water.
For example, sodium chloride melts at approximately 801°C due to strong electrostatic forces within its ionic lattice.
Covalent compounds:
- Often have lower melting points.
- May not conduct electricity.
- Can be gases, liquids, or soft solids.
This distinction helps predict chemical behavior, reactivity patterns, physical properties, and material stability.
Real-World Examples of Ionic Bonds
- Sodium chloride (NaCl) – table salt
- Magnesium oxide (MgO)
- Calcium fluoride (CaF₂)
These substances exist as extended ionic lattices rather than discrete molecules.
These compounds form crystal lattices due to strong ionic attraction.
Common Misconceptions
Misconception 1: Ionic bonds share electrons unequally.
Ionic bonds do not share electrons at all. They involve full transfer.
Misconception 2: Ionic bonds are always stronger than covalent bonds.
Bond strength depends on the environment and measurement method.
For example, the carbon–carbon (C–C) bond has a bond dissociation energy of about 348 kJ/mol, which can be comparable to or stronger than some ionic interactions in solution.
Misconception 3: All bonds follow the octet rule perfectly.
Some molecules (like expanded octet species) do not strictly follow the octet rule.
One-Line Summary
Ionic bonds form through electron transfer, while covalent bonds form through electron sharing.
FAQs
1. Do ionic bonds share electrons equally or unequally?
Ionic bonds do not share electrons. Electrons are transferred from one atom to another.
2. What happens to electrons in an ionic bond?
One atom loses electrons, and another gains them, forming oppositely charged ions.
3. Which bond is stronger: ionic or covalent?
It depends on the compound and environment. Neither type is universally stronger.
4. What shares electrons unequally?
Polar covalent bonds share electrons unequally due to electronegativity differences.
5. Why do metals form ionic bonds easily?
Metals have low ionization energy and can easily lose valence electrons.
The Bottom Line
Ionic bonds do not share electrons.
They form when electrons are transferred from one atom to another, creating oppositely charged ions that attract each other.
Electron sharing occurs in covalent bonds.
Understanding this difference is essential for predicting chemical properties, reactivity patterns, material behavior, and molecular structure.
These bonding principles are central to the study of chemistry and help explain material properties at the atomic level.

The Science Editorial Team creates clear, accurate, and student-friendly explanations of scientific concepts across biology, chemistry, physics, taxonomy, anatomy, and related life sciences. Content is written for educational and informational purposes using standard academic references and reliable sources. Read more about the → Science Editorial Team