Quick Answer: Chemistry is essential in nursing because safe patient care depends on understanding medications, electrolyte balance, acid–base regulation, IV solutions, and laboratory values. Nurses apply chemical principles daily when administering drugs, monitoring blood chemistry, and responding to emergencies.
Without chemistry knowledge, safe nursing practice would be severely limited.
Modern healthcare standards emphasize medication safety and biochemical monitoring as core nursing competencies.
This explains why chemistry is a required subject in most nursing programs worldwide.
The broader role of chemistry in society is discussed in Importance of Chemistry in Daily Life.
Nursing Is Applied Chemistry
Nursing practice relies heavily on applied chemistry principles in clinical care.
The human body is a chemical system. The practical application of chemical science is explained in Applied Chemistry.
Every second, chemical reactions control:
- Breathing
- Digestion
- Hormone regulation
- Nerve transmission
- Muscle contraction
- Blood clotting
Nurses work directly with these processes.
Understanding chemistry allows nurses to interpret what is happening inside the body — and act correctly.
In clinical practice, nurses do not simply follow instructions — they make rapid, informed decisions based on patient chemistry. Changes in blood pressure, oxygen saturation, glucose levels, or electrolyte balance often reflect underlying chemical shifts in the body. Understanding these shifts improves patient safety and reduces medication errors.
Why Do Nurses Study Chemistry?
Nursing programs include chemistry because it builds the foundation for:
- Pharmacology
- Biochemistry
- Physiology
- Pathophysiology
- Clinical medication safety
Chemistry helps nurses move from memorization to understanding.
Role of Chemistry in Nursing Practice
The role of chemistry in nursing extends beyond textbooks. It directly influences medication administration, fluid therapy, laboratory monitoring, and emergency response. In clinical settings, nurses rely on chemical principles to ensure accurate treatment and prevent complications.
Key Applications of Chemistry in Nursing
Chemistry in Pharmacology
This is the most important area.
Nurses must understand:
- Drug composition
- Drug interactions
- Dosage calculations
- Metabolism in the liver
- Drug excretion through kidneys
- Side effects and toxicity
Drug action occurs at the molecular level. Medications bind to receptors, alter enzyme activity, or modify ion channels. Nurses must understand half-life, therapeutic index, metabolism pathways (such as hepatic enzyme systems), and elimination processes to prevent toxicity or underdosing.
For example:
- NSAIDs combined with anticoagulants increase bleeding risk.
- Alcohol mixed with sedatives slows the central nervous system.
Without chemistry knowledge, medication errors increase.
Safe drug administration depends on chemistry.
Chemistry in IV Fluids and Electrolytes
IV solutions are chemical solutions.
Examples include:
- Normal saline (0.9% NaCl)
- Ringer’s lactate
- Dextrose solutions
Nurses must understand:
- Osmosis
- Electrolyte balance (Na⁺, K⁺, Ca²⁺). If you may want to check the behavior of charged particles in the body, see Difference Between Cations and Anions.
- Fluid shifts
- Dehydration and overhydration
An incorrect electrolyte level can cause:
- Cardiac arrhythmia
- Muscle weakness
- Confusion
- Seizures
For example, severe hyperkalemia can trigger fatal cardiac arrhythmias, while hyponatremia may lead to cerebral edema and confusion. Nurses must recognize these biochemical imbalances early and respond appropriately.
Chemistry knowledge helps prevent life-threatening complications.
Acid–Base Balance and pH
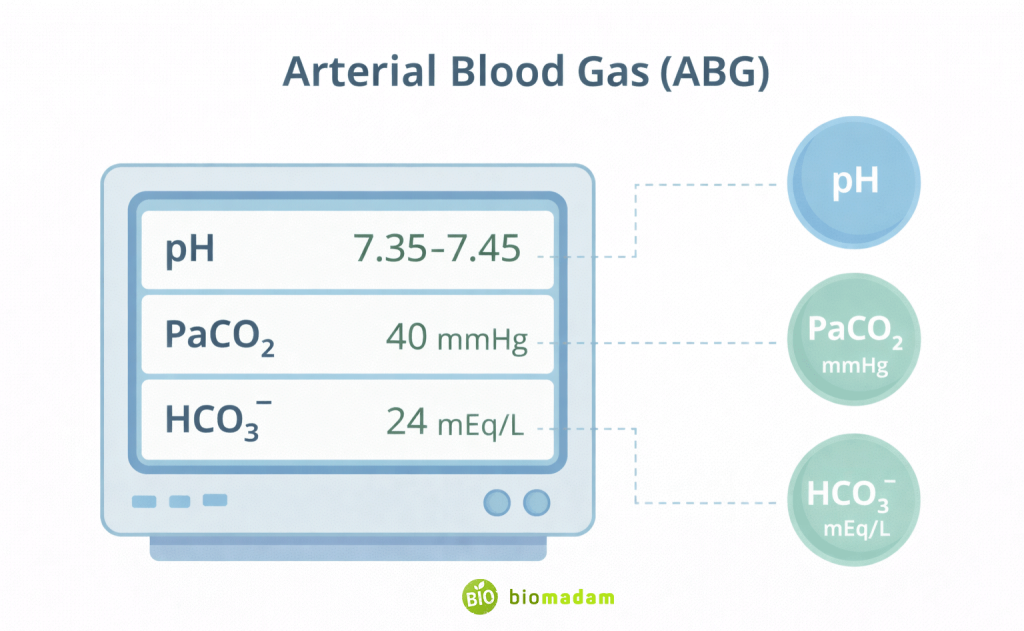
Blood pH is tightly regulated between 7.35 and 7.45. Even small deviations can become life-threatening.
Nurses monitor four primary acid–base disorders:
- Respiratory acidosis
- Respiratory alkalosis
- Metabolic acidosis
- Metabolic alkalosis
Arterial blood gas (ABG) analysis measures:
- pH – overall acidity or alkalinity
- PaCO₂ – respiratory component
- HCO₃⁻ – metabolic component
By evaluating these values together, nurses determine whether the imbalance is respiratory or metabolic and whether compensation is occurring.
For example, elevated PaCO₂ indicates respiratory acidosis, commonly seen in respiratory failure. Reduced HCO₃⁻ suggests metabolic acidosis, which may occur in sepsis, kidney failure, or diabetic ketoacidosis.
Understanding hydrogen ion concentration (H⁺), carbon dioxide balance, and bicarbonate buffering allows nurses to interpret ABG results accurately and respond promptly in critical care settings.
Acid–base disturbances require immediate attention, and chemistry knowledge enables safe and timely intervention.
Chemistry in Lab Test Interpretation
Many lab tests are chemical measurements.
Examples:
- Blood glucose
- Creatinine
- Electrolytes
- Hemoglobin
- Liver enzymes
Nurses must understand what abnormal values mean.
For example:
High potassium (hyperkalemia) can stop the heart.
Chemistry knowledge turns numbers into clinical decisions.
Biochemistry of the Human Body
Biochemistry explains:
- Carbohydrate metabolism
- Protein synthesis
- Lipid breakdown
- Enzyme activity
- Hormonal signaling
Diabetes management depends on understanding glucose chemistry.
Nutrition planning depends on metabolic chemistry.
Nurses use this knowledge daily.
Drug–Food and Drug–Beverage Interactions
Chemistry explains:
- Why grapefruit juice affects certain drugs
- Why iron tablets should not be taken with milk
- Why alcohol enhances sedative effects
Nurses educate patients using this chemical understanding.
Patient safety improves with proper knowledge.
Why Nursing Students Struggle With Chemistry
Common challenges include:
- Fear of science subjects
- Weak math foundation
- Difficulty connecting theory to practice
The solution is application-based learning. When students see how chemistry saves lives, it becomes meaningful.
The difficulty often arises when chemistry is taught without clinical connection. When linked directly to IV fluids, emergency drug administration, and patient monitoring, chemistry becomes practical rather than abstract. Nursing education increasingly emphasizes applied chemistry for this reason.
Common Misconception
Some students believe chemistry is only theoretical.
In reality:
- Every medication given
- Every IV started
- Every lab report reviewed
- Every emergency handled
Involves chemistry.
Chemistry Subjects Required in Nursing Programs
Most accredited nursing programs include:
- General Chemistry
- Organic Chemistry
- Biochemistry
- Pharmacology
- Pathophysiology
These subjects fall under the broader structure outlined in 5 Branches of Chemistry.
These subjects prepare nurses to understand molecular interactions, metabolic pathways, and drug mechanisms essential in clinical care.
Chemistry and Patient Safety
Chemistry helps nurses understand medications, body processes, and clinical lab results to ensure safe and effective patient care.
Medication errors, improper IV administration, and failure to recognize abnormal lab values are major contributors to patient harm. A strong foundation in chemistry reduces these risks. By understanding chemical interactions, concentration calculations, and physiological reactions, nurses improve treatment accuracy and patient outcomes.
FAQs
Why is chemistry important in nursing?
Chemistry is important in nursing because it explains how medications work, how IV fluids affect electrolyte balance, how lab values reflect body chemistry, and how drug interactions occur. This knowledge ensures safe and effective patient care.
Do nurses really use chemistry in hospitals?
Yes. Nurses use chemistry daily when administering drugs, monitoring electrolytes, and interpreting lab tests.
Is pharmacology part of chemistry?
Yes. Pharmacology is based on chemical interactions between drugs and the body.
Is chemistry difficult for nursing students?
It can be challenging, but understanding its clinical relevance makes it easier and more practical.
The Bottom Line
In modern healthcare systems, medication safety and biochemical monitoring are core nursing responsibilities, and both depend heavily on applied chemistry knowledge.
Nursing is both compassionate and scientific. Chemistry provides the molecular understanding that supports safe medication administration, critical decision-making, and high-quality patient care across all healthcare settings.

The Science Editorial Team creates clear, accurate, and student-friendly explanations of scientific concepts across biology, chemistry, physics, taxonomy, anatomy, and related life sciences. Content is written for educational and informational purposes using standard academic references and reliable sources. Read more about the → Science Editorial Team

Pls can u send nursing PTS past question papper
Please can you help me with pts pass question for midwifery
What is the importance to nursing