Quick Answer: Ion exchange chromatography has several disadvantages, including slow equilibration, sensitivity to pH changes, limited compatibility with organic solvents, column inconsistency, and high operational costs.
Although it is powerful for separating charged molecules, it requires careful control of experimental conditions and may not always provide structural information.
Ion exchange chromatography is widely used in analytical chemistry, biochemistry, and environmental analysis. However, like all separation techniques, it has practical and technical limitations that must understand before applying it in laboratory experiments.
What This Article Covers
- Basic principle of ion exchange chromatography
- Major disadvantages explained clearly
- Practical laboratory limitations
- Comparison with other chromatographic methods
- Why these limitations matter in experiments
Core Concept: How Ion Exchange Chromatography Works
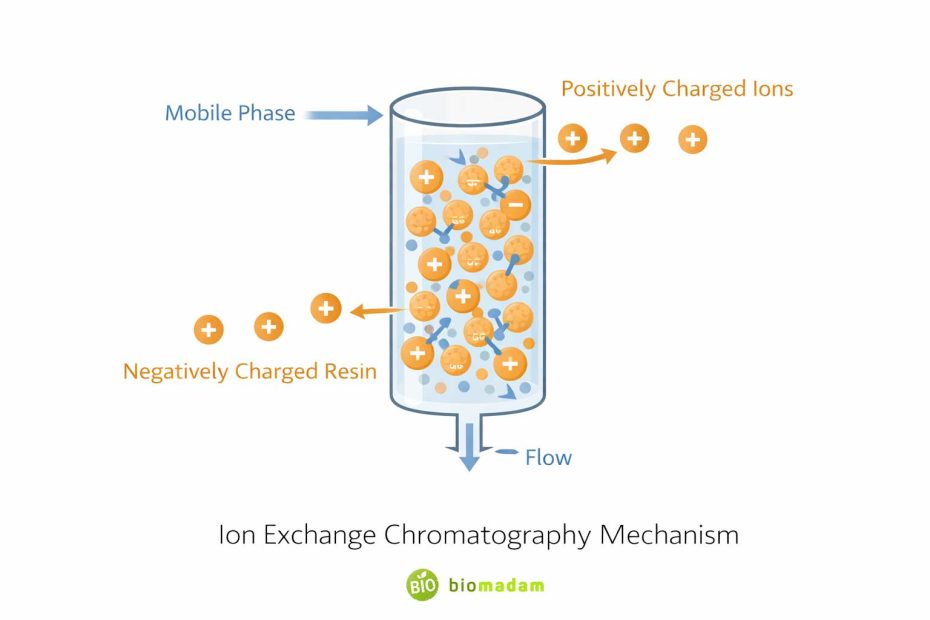
Ion exchange chromatography separates molecules based on their charge.
The general separation mechanism is explained in Principle of Chromatography.
A charged stationary phase (resin) binds oppositely charged molecules from a mixture. The bound molecules are later released (eluted) by changing salt concentration or pH.
There are two main types:
- Cation exchange chromatography – separates positively charged molecules
- Anion exchange chromatography – separates negatively charged molecules
Although the principle is simple, practical limitations arise during real experiments.
The behavior of charged species is explained in Difference Between Cations and Anions.
Why It Matters
Ion exchange chromatography is widely used for:
- Protein purification
- Amino acid separation
- Water analysis
- Pharmaceutical research
However, understanding its disadvantages helps students choose the correct separation technique and avoid experimental errors.
For example, during protein purification, a small change in pH can alter a protein’s net charge. If the protein loses its charge, it may fail to bind to the column entirely, resulting in poor separation or sample loss.
Major Disadvantages of Ion Exchange Chromatography
The following explain the main limitations and drawbacks of ion exchange chromatography in practical laboratory use.
Strong Dependence on pH
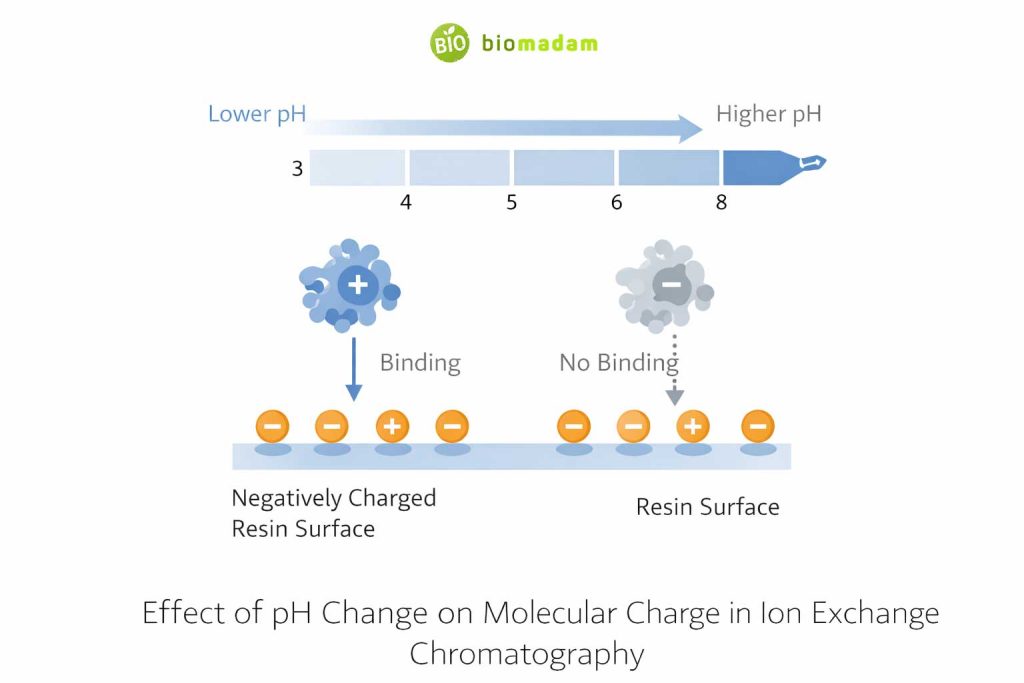
The separation depends heavily on the pH of the mobile phase.
If pH changes slightly:
- The charge on the molecule may change. Molecular charge behavior is discussed in How Are Cations Formed?
- Binding strength may decrease or increase.
- Separation may fail.
This makes method optimization time-consuming.
Slow Equilibration and Long Run Times
Before sample injection, the column must be equilibrated with buffer solution.
After each run:
- The column may require regeneration.
- Additional washing steps are needed.
This increases total experimental time.
Limited Information About Molecular Structure
Ion exchange chromatography separates molecules based on charge only.
It does not:
- Provide structural identification
- Confirm molecular weight
- Identify functional groups
Additional detectors (UV, MS, conductivity) are required.
Diffusion and Pore Size Limitations
Resin beads contain pores.
Surface interaction processes are explained in Difference Between Absorption and Adsorption.
Large biomolecules such as proteins may:
- Diffuse slowly into pores
- Experience steric hindrance
- Show reduced separation efficiency
Improper pore size selection reduces resolution.
Inconsistency Between Columns
Different manufacturers produce columns with:
- Different resin materials
- Different functional groups
- Different particle sizes
This may cause:
- Variation in retention times
- Reduced reproducibility
- Difficulty in method transfer
High Cost of Columns and Maintenance
Ion exchange columns are often expensive.
Additional costs include:
- Buffer preparation
- Salt gradients
- Regeneration chemicals
- Maintenance of system components
Compared to some simpler techniques, operating costs may be higher.
Sensitivity to Organic Solvents
Ion exchange resins are generally designed for aqueous systems.
High concentrations of organic solvents may:
- Damage resin structure
- Reduce binding efficiency
- Shorten column life
This limits versatility.
Complex Buffer Preparation
Ion exchange chromatography requires carefully prepared buffer systems.
Incorrect pH or ionic strength can significantly affect separation efficiency.
Gradient elution methods often require multiple buffer solutions, increasing preparation time and experimental complexity.
Column Fouling and Resin Degradation
Biological samples may contain impurities that bind irreversibly to the resin.
Over time, this can reduce column performance, decrease resolution, and shorten column lifespan.
Regular regeneration and maintenance are required to preserve efficiency.
Comparison With Reverse Phase Chromatography
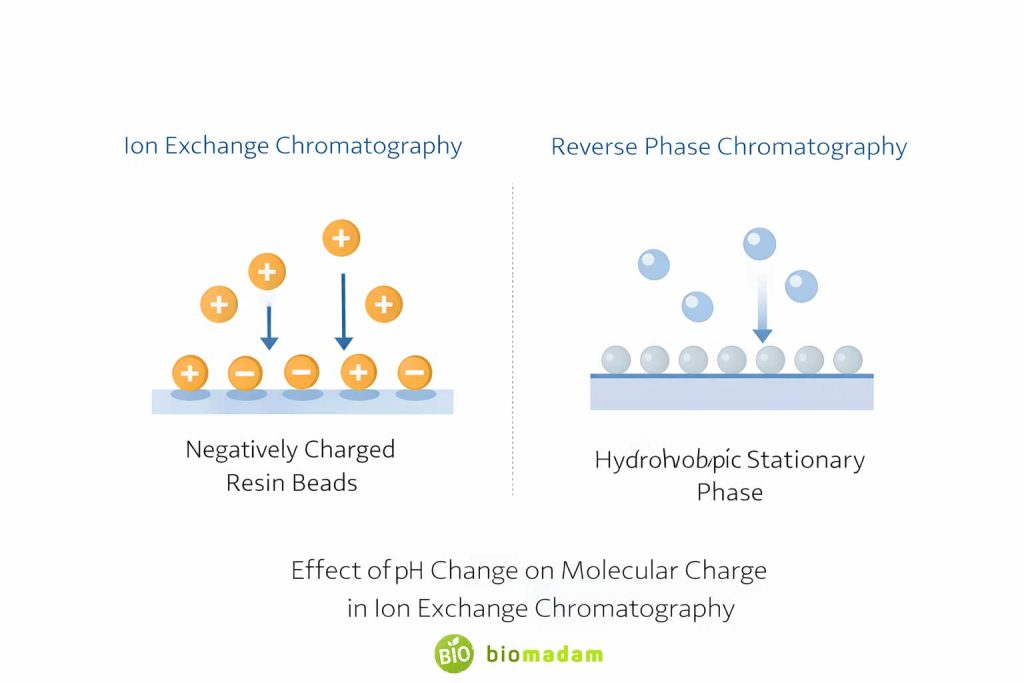
Ion exchange chromatography is one of the techniques discussed under Types of Column Chromatography.
| Feature | Ion Exchange Chromatography | Reverse Phase Chromatography |
| Basis of Separation | Charge | Hydrophobicity |
| pH Sensitivity | High | Moderate |
| Organic Solvent Compatibility | Limited | High |
| Structural Information | Requires detector | Requires detector |
| Method Development Difficulty | Moderate to High | Moderate |
| Column Durability | Sensitive to fouling | Generally more stable |
| Typical Use | Charged biomolecules | Small organic molecules |
Reverse phase chromatography is often more stable and widely used, but it cannot separate molecules purely based on charge.
Common Misconceptions
Misconception 1: Ion exchange chromatography gives complete molecular information.
It only separates based on charge.
Misconception 2: It works for all types of molecules.
It works best for charged or polar compounds.
Misconception 3: It is always superior to reverse phase chromatography.
The best method depends on the sample type.
When Ion Exchange Chromatography Is Not Ideal
Ion exchange chromatography is not suitable for neutral, non-polar compounds that lack charge.
In such cases, techniques like reverse phase chromatography or size exclusion chromatography may provide better separation results.
One-Line Summary
Ion exchange chromatography is powerful for separating charged molecules, but it requires strict pH control, careful column selection, and additional analytical tools.
FAQs
1. Why is pH important in ion exchange chromatography?
Because molecular charge changes with pH, affecting binding strength.
2. Is ion exchange chromatography expensive?
Yes, columns and maintenance costs can be high.
3. Can it separate neutral molecules?
No. It mainly separates charged species.
4. Why is equilibration time long?
The resin must reach ionic balance before accurate separation.
5. When should ion exchange chromatography be used?
When separating ions, proteins, nucleic acids, or charged biomolecules.
6. What are the limitations of ion exchange chromatography in protein purification?
This targets very specific search queries.
The Bottom Line
Ion exchange chromatography is a valuable analytical tool, especially for charged biomolecules. However, it has limitations related to pH sensitivity, cost, reproducibility, and structural identification. Understanding these disadvantages helps students choose the correct technique for their experiments.

The Science Editorial Team creates clear, accurate, and student-friendly explanations of scientific concepts across biology, chemistry, physics, taxonomy, anatomy, and related life sciences. Content is written for educational and informational purposes using standard academic references and reliable sources. Read more about the → Science Editorial Team