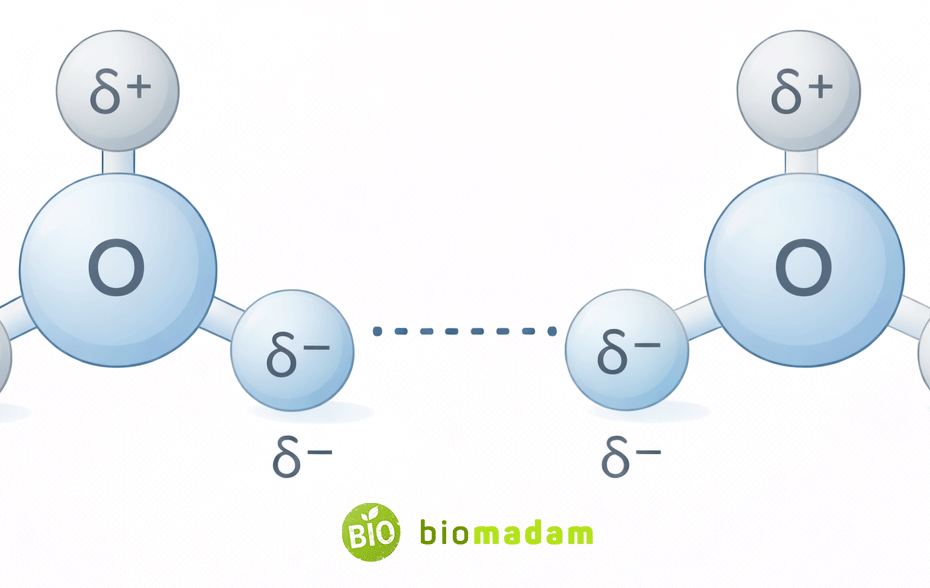
Quick Answer: No, hydrogen bonds do not share electrons like covalent bonds.
Instead, a hydrogen bond is a weak electrostatic attraction between a hydrogen atom that is already covalently bonded to a highly electronegative atom (such as oxygen, nitrogen, or fluorine) and another nearby electronegative atom.
Hydrogen bonding occurs because of partial charges created by polar covalent bonds, not because electrons are shared between molecules.
Core Concept: What Is a Hydrogen Bond?
A hydrogen bond is a special type of dipole–dipole interaction.
It occurs when a hydrogen atom bonded to a highly electronegative atom interacts with another electronegative atom nearby.
Typical hydrogen-bond donors include:
- O–H (oxygen–hydrogen)
- N–H (nitrogen–hydrogen)
- F–H (fluorine–hydrogen)
These bonds create partial charges:
- Hydrogen becomes partially positive (δ⁺)
- The electronegative atom becomes partially negative (δ⁻)
The positive hydrogen then attracts another electronegative atom with lone electron pairs.
This attraction forms a hydrogen bond.
Hydrogen bonds are:
- Stronger than van der Waals forces
- Weaker than covalent or ionic bonds
A hydrogen bond typically forms when three atoms align in the pattern:
X–H···Y, where:
- X is an electronegative atom (O, N, or F) bonded to hydrogen
- H is the hydrogen atom
- Y is another electronegative atom with lone electron pairs
This geometry allows strong electrostatic attraction between molecules.
Do Hydrogen Bonds Share Electrons?
No.
Hydrogen bonds do not share electrons between atoms.
Instead, they form through electrostatic attraction between a partially positive hydrogen atom and a nearby electronegative atom with lone electron pairs.
In hydrogen bonding:
- The hydrogen atom is already covalently bonded to another atom.
- That hydrogen carries a partial positive charge.
- A nearby electronegative atom with lone electron pairs attracts that hydrogen.
Because of this attraction:
- Molecules are held together temporarily.
- No new electron-sharing bond is formed.
This is why hydrogen bonds are classified as intermolecular forces, not true chemical bonds.
Types of Hydrogen Bonds
Hydrogen bonding occurs in two main forms.
Intramolecular Hydrogen Bonding
Intramolecular hydrogen bonding occurs within a single molecule.
The hydrogen donor and acceptor are part of the same molecule, forming an internal interaction.
Example:
- Certain organic molecules such as salicylic acid or ethylene glycol can form internal hydrogen bonds.
These bonds often influence the shape and stability of molecules.
Intermolecular Hydrogen Bonding
Intermolecular hydrogen bonding occurs between different molecules.
The hydrogen atom of one molecule interacts with an electronegative atom of another molecule.
Common examples:
- Water (H₂O)
- Ammonia (NH₃)
- Hydrogen fluoride (HF)
These interactions strongly affect physical properties like boiling point and solubility.
Common Examples of Hydrogen Bonding
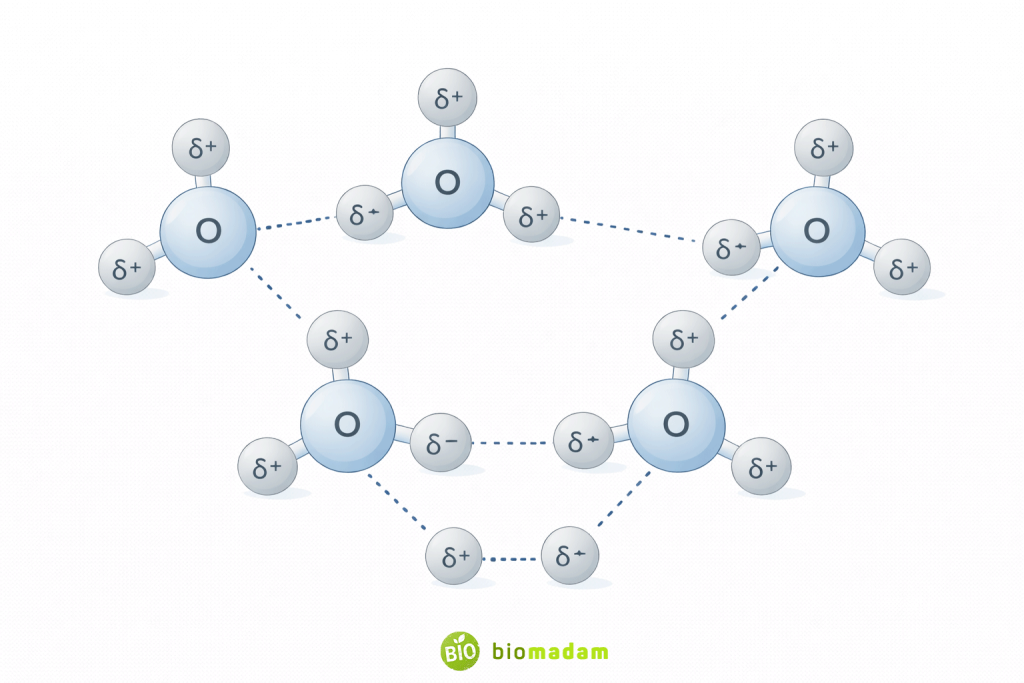
Hydrogen bonding appears in many chemical and biological systems.
Water (H₂O)
Each water molecule can form up to four hydrogen bonds — two through its hydrogen atoms and two through the lone electron pairs on oxygen.
This network explains many unusual properties of water.
Hydrogen Fluoride (HF)
Fluorine is extremely electronegative, producing very strong hydrogen bonds.
Ammonia (NH₃)
Nitrogen forms hydrogen bonds through its lone pair electrons.
Alcohols
Alcohols contain –OH groups, which readily participate in hydrogen bonding.
Biological Molecules
Hydrogen bonds play a major role in:
- DNA base pairing (A–T and G–C interactions)
- Protein folding and stability
- Cellulose fiber structure in plants
Properties Caused by Hydrogen Bonding
Hydrogen bonds typically have bond energies ranging from 5 to 40 kJ/mol, depending on the molecules involved and the surrounding environment.
Hydrogen bonding significantly influences physical and chemical properties, as discussed in Difference Between Physical and Chemical Properties.
Higher Boiling Points
Compounds with hydrogen bonding often have higher boiling points than similar molecules without it.
Example: Water boils at 100 °C, much higher than comparable molecules.
Increased Surface Tension
Hydrogen bonds create strong molecular attraction, giving water high surface tension.
Higher Viscosity
Liquids with hydrogen bonding resist flow more strongly.
Examples include glycerol and alcohols.
Ice Has Lower Density Than Water
Hydrogen bonds create an open lattice structure in ice.
This structure creates an open hexagonal lattice that increases the distance between water molecules. As a result, ice becomes less dense than liquid water and floats.
Hydrogen Bonds vs Covalent Bonds
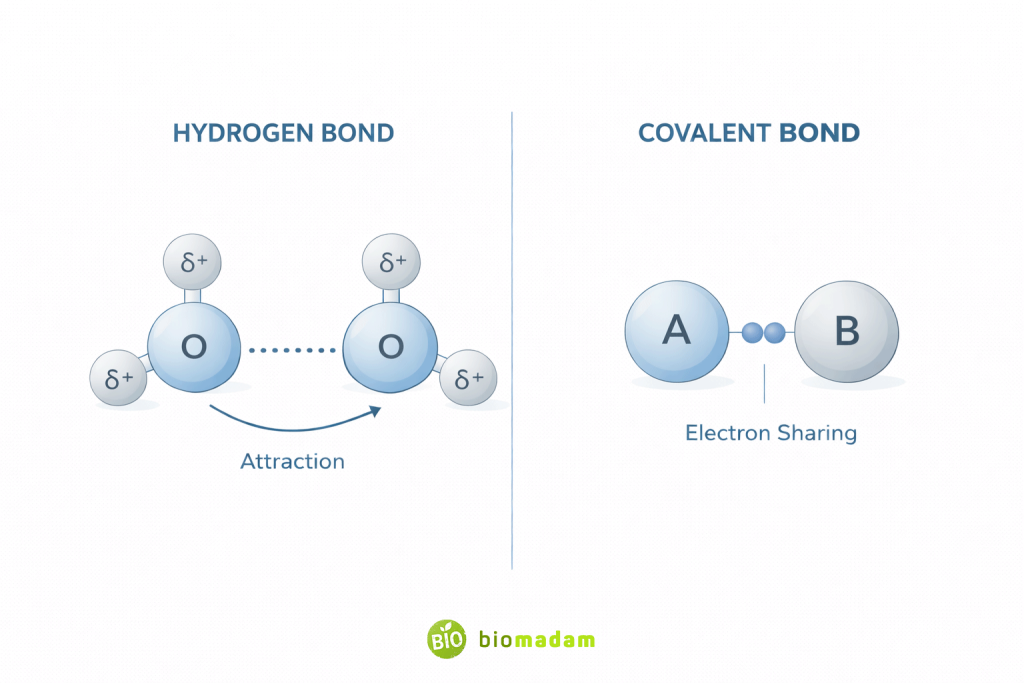
| Feature | Hydrogen Bond | Covalent Bond |
| Electron Sharing | No | Yes |
| Bond Type | Intermolecular attraction | True chemical bond |
| Bond Strength | Weak–moderate (5–40 kJ/mol) | Strong (100–1000 kJ/mol) |
| Formation | Partial charge attraction | Shared electron pair |
| Example | H₂O hydrogen bonding | O–H bond in water |
A detailed comparison is explained in Difference Between Hydrogen Bond and Covalent Bond.
Why Hydrogen Bonding Matters
Hydrogen bonding plays a crucial role in both chemistry and biology.
It helps explain:
- The structure of DNA
- Protein folding and stability
- The properties of water
- Solubility of many organic compounds
Without hydrogen bonding, many biological systems would not function properly.
One-Line Summary
Hydrogen bonds do not share electrons; they form through electrostatic attraction between a partially positive hydrogen atom and a nearby electronegative atom.
FAQs
Do hydrogen bonds share electrons?
No. Hydrogen bonds do not involve electron sharing. They form through electrostatic attraction between partial charges.
Do ionic bonds share electrons?
No. Ionic bonds do not share electrons. Instead, electrons are transferred from one atom to another, forming oppositely charged ions that attract each other through electrostatic forces. This process is explained in Do Ionic Bonds Share Electrons?
Are hydrogen bonds stronger than covalent bonds?
No. Hydrogen bonds are weaker than covalent or ionic bonds, which are explained in Difference between Ionic, Covalent and Metallic Bonds.
Where do hydrogen bonds occur?
They occur in molecules containing O–H, N–H, or F–H bonds, such as water, alcohols, and ammonia.
Why is hydrogen bonding important in biology?
Hydrogen bonds stabilize DNA structure, protein folding, and molecular interactions in cells.
Do hydrogen bonds exist within molecules?
Yes. Intramolecular hydrogen bonds can occur within a single molecule when donor and acceptor groups are close together.
The Bottom Line
Hydrogen bonds do not share electrons. They form through electrostatic attraction between a partially positive hydrogen atom and a nearby electronegative atom, influencing the properties of water, biomolecules, and many chemical systems.
They are weak electrostatic attractions that occur when a partially positive hydrogen atom interacts with a nearby electronegative atom. Despite being weaker than true chemical bonds, hydrogen bonding plays a critical role in determining the structure, properties, and biological functions of many molecules.

The Science Editorial Team creates clear, accurate, and student-friendly explanations of scientific concepts across biology, chemistry, physics, taxonomy, anatomy, and related life sciences. Content is written for educational and informational purposes using standard academic references and reliable sources. Read more about the → Science Editorial Team