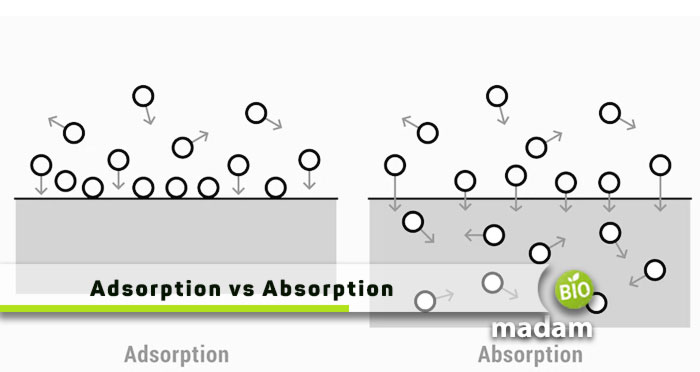
Adsorption and absorption are among the important phenomena in biology and chemistry besides diffusion and effusion, and osmosis. While the terms seem quite similar to each other, they differ slightly. Absorption is the entry of any substance into the bulk phase of a solid or liquid. On the other hand, adsorption is the attachment of compounds to molecules.
Let’s tell you everything about the differences between absorption and adsorption.
Comparison Table
| Characteristics | Absorption | Adsorption |
| Definition | Bulk movement of molecules | Attachment of molecules to surface |
| Phenomenon | Bulk phenomenon | Surface phenomenon |
| Heat Exchange | Endothermic | Exothermic |
| Temperature | No Effect | Inversely proportional |
| Occurrence | More often | Seldom occurrence |
| Rate of Reaction | Steady | Increase after achieving equilibrium |
| Molecular Interaction | More | Less |
| Concentration | Steady | Changes |
What is Absorption?
Absorption is when a aqueous or liquid is soaked up through a substance like a sponge, cloth, or filter paper. Simply put, absorption refers to a phenomenon an absorbent material absorbs a liquid. In biology, absorption has vast applications in tissues and organs that absorb materials through different types of diffusion and osmosis. The skin also allows the absorption of materials through the surface. Moreover, inhalation through the right and left lungs and ingestion are also categorized as absorption. Considering that substances either exhibit physical or chemical changes, absorption also varies.
Types of Absorption
There are two types of absorption, i.e., chemical absorption and physical absorption.
Chemical Absorption
Chemical absorption refers to the absorption of chemicals in one state by chemicals in another state of matter. It generally occurs by the chemical reaction between the absorbing and absorbed material. Chemical absorption is critical to many industries, including occupational safety controls.
Physical Absorption
The other type of absorption is physical absorption. Physical absorption, also known as non-reactive absorption, refers to the reaction when oxygen in the atmosphere dissolves in water. Thus, no reaction occurs during the absorption process. It takes place when multiple elements in the resulting substance interact with each other.
Examples
- Cold storage
- Refrigerators
- Chillers for cooling space
- Turbine inlet cooling
- Amine washing technology
- Absorption of carbon dioxide from the gasses of a greenhouse
- Absorption of food from the small and large intestines
- Production of ice
What is Adsorption?
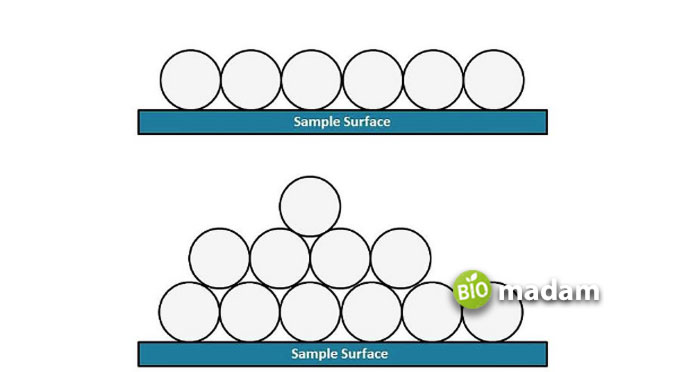
Adsorption refers to when atoms, ions, or molecules gather on the surface area. These molecules come in contact with the surface area through chemical bonds. This interaction allows molecules to adsorb in the area of a surface. It depends on the size and portion of the surface area. The larger the surface area will be, the more molecules will interact with the surface area and then adsorb it. That surface area consists of different atoms that are in contact with air.
Types of Adsorption
Chemisorption
Chemisorption is the process by which gaseous molecules come in contact with the surface area through a chemical bond. It is also called chemical adsorption and takes place in adsorbed substances held together by chemical bonds. It is a highly specific process and occurs only when the adsorbate and adsorbent have a chemical bonding. Considering that this type of adsorption alters the chemical properties, it is irreversible and has high pressure. It also depends on the surface area and is directly proportional. Thus, a material’s chemisorption also increases with a larger surface area.
Physisorption
Physisorption occurs when gaseous molecules contact the surface area through repulsive forces between particles and electrostatic forces. It is also known as physical adsorption. Physisorption is an exothermic process and releases low enthalpy. The gasses typically accumulate on the solid surface resulting from weak Van der Waals forces. This process depends on the adsorbent’s surface area. The extent of adsorption increases with the increase in surface area. Metals with a large surface area are thought to be better adsorbents.
Examples
- Air conditions
- Water purification
- The development process of non-stick coating
- Inside gas masks to adsorb poisonous gasses
- Silica gels to adsorb moisture
- Charcoal to separate noble gasses
Difference Between Absorption and Adsorption
Definition
Absorption
The concept of absorbing a liquid or solid with the help of a substance is known as absorption.
Adsorption
On the other hand, different molecules getting together and sticking on the surface is known as adsorption.
Phenomenon
Absorption
It is a bulk phenomenon that is when molecules, cations, or anions enter a material in bulk condition. Such as soaking the whole liquid spread on the ground. In this condition, different ions and molecules will come in contact with a substance in the bulk phase.
Adsorption
On the contrary, adsorption works on the surface area. Such as interaction and the stickiness of molecules with the surface area.
Heat Exchange
Absorption
Absorption is clearly an endothermic process.
Adsorption
Whereas, adsorption is an exothermic process.
Temperature
Absorption
There is no effect of temperature on absorption.
Adsorption
However, adsorption may be affected by low temperatures. Adsorption decreases with the increase in temperature.
Rate of Reaction
Absorption
There is no drastic change in the rate of reaction of absorption. There is a uniform rate of reaction in the case of absorption.
Adsorption
At the same time, the reaction rate of adsorption increases gradually over time and then reaches the state of equilibrium at one point.
Concentration
Absorption
As there is no significant change in the rate of absorption reaction, the concentration remains constant throughout the whole interaction.
Adsorption
But, the concentration level at the bottom of adsorption is different than at the beginning.
The Bottom Line
Absorption and adsorption are widely spread phenomenon in our communities to maintain an ecosystem that occur in different forms. Absorption is common in daily life, such as cleaning water spills with a tissue or cloth. On the other hand, adsorption also plays a major role in human physiology besides environmental changes. Silica gels are a critical example of adsorption in our daily life. One of the basic differences between absorption and adsorption is an energy exchange. Absorption is an endothermic process, while adsorption is an exothermic process.
FAQs
What is the main difference between adsorption and absorption?
The main difference between adsorption and absorption is the behavior of molecules toward the other substance. Molecules in absorption enter the bulk phase while they stick to the surface of the material in adsorption.
What is an example of adsorption and absorption?
The absorption of water into an absorbing material is an example of absorption. On the contrary, the attachment of water vapors onto the silica gel to avoid dampening represents adsorption.
What is used in absorption?
Essentially any absorbing material can be used in absorption. However, absorption systems typically utilize lithium bromide and water, where the lithium bromide acts as the absorber.

The Biomadam Content Team develops and manages educational and informational articles published on Biomadam. Content is prepared using standard reference materials and follows internal editorial guidelines to ensure clarity and consistency.