Quick Answer: Organic compounds are widely used in daily life as food components, medicines, fuels, household products, and industrial materials. Common organic compounds such as carbohydrates, fats, hydrocarbons, alcohols, and organic acids play essential roles in energy production, nutrition, healthcare, transportation, and manufacturing.
What Are Organic Compounds?
Organic compounds are chemical substances that primarily contain carbon and hydrogen, often along with elements such as oxygen, nitrogen, sulfur, or phosphorus. Most organic compounds form the basis of life and are central to biological and industrial processes.
Earlier, organic compounds were believed to originate only from living organisms. However, modern chemistry defines organic compounds based on their carbon-based structure, not their origin.
Exceptions: Certain carbon-containing substances such as carbonates, cyanides, and carbides are considered inorganic.
The branch of chemistry that studies these compounds, their structure, reactions, and properties is called organic chemistry.
Historically, scientists believed that organic compounds could only be produced by living organisms because of a supposed “vital force.” This misconception was corrected in 1828 when Friedrich Wöhler synthesized urea from inorganic substances, proving that organic compounds could be created in laboratories. This discovery laid the foundation for modern organic chemistry and changed how scientists define organic compounds today.
Key Characteristics of Organic Compounds
Organic compounds show some common features:
- Carbon atoms are covalently bonded, giving structural stability
- Generally have low melting and boiling points
- Mostly non-polar or weakly polar
- Poor conductors of electricity
- Often combustible
These characteristics explain why organic compounds are so well suited for life and everyday applications. Covalent bonding allows carbon atoms to form stable yet flexible structures, enabling the formation of complex molecules such as sugars, fats, and proteins. Their relatively low melting points and combustibility make many organic compounds useful as fuels, food components, and industrial materials.
Based on their chemical properties and biological compatibility, organic compounds find extensive applications in everyday activities.
Uses of Organic Compounds in Daily Life
Organic compounds are present in almost every aspect of everyday life. Some major categories and examples are discussed below.
1. Organic Compounds in Food and Nutrition
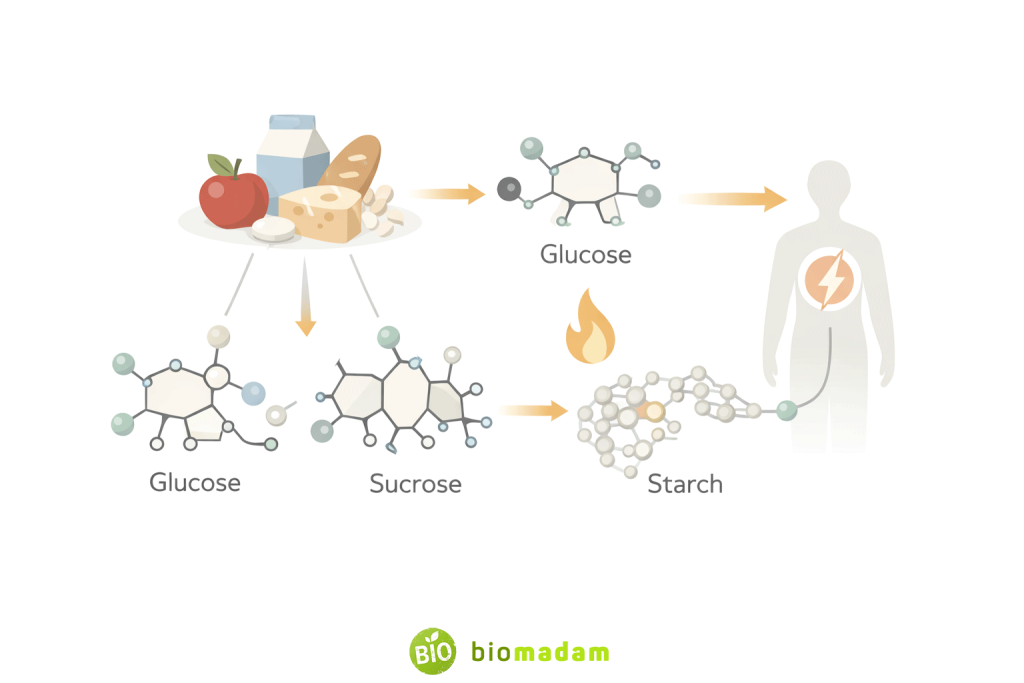
Carbohydrates (Glucose, Sucrose, Starch)
Structural differences among simple sugars are discussed in the Difference Between Aldose and Ketose.
- Glucose is the primary source of energy for cells and circulates in blood
- Sucrose (table sugar) is widely used as a sweetener in foods and beverages
- Starch is the main storage carbohydrate in plants and a major dietary energy source
Uses:
- Energy production
- Food thickening (soups, sauces)
- Infant formulas and sports drinks
- Food preservation (jams, jellies)
In the human body, carbohydrates are digested into simpler sugars such as glucose, which is absorbed into the bloodstream and used by cells for immediate energy. Excess glucose is stored in the liver and muscles as glycogen or converted into fats for long-term energy storage. This ability to provide both quick and stored energy makes carbohydrates essential for daily metabolic activities.
2. Organic Compounds in Medicine
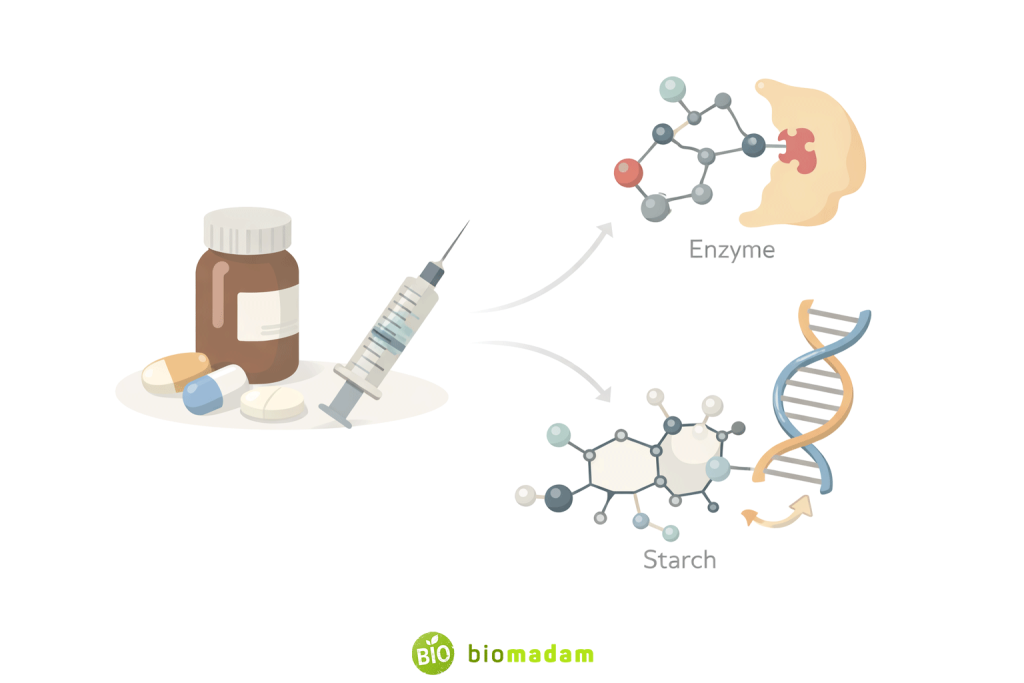
Glucose
- Administered intravenously to patients who cannot eat
- Used in treating hypoglycemia
Acetic Acid
- Used as an antiseptic and antimicrobial agent
- Helps in medical screening procedures
- Used in pharmaceuticals and chemical synthesis
Organic Drugs
Many medicines such as aspirin, paracetamol, antibiotics, and vitamins are organic compounds.
Most medicines are organic compounds because carbon-based molecules can interact effectively with biological systems. Their structural similarity to natural biomolecules allows drugs to bind precisely to enzymes, receptors, or DNA, making treatments more specific and efficient. This is why painkillers, antibiotics, and vitamins are predominantly organic in nature.
3. Organic Compounds in Household Use
Fats and Oils
- Provide stored energy
- Help absorb fat-soluble vitamins (A, D, E, K)
- Used in cooking, cosmetics, and soaps
Organic Acids
- Acetic acid in vinegar
- Citric acid in food preservation and flavoring
Organic compounds are widely used in household products for cleaning, hygiene, and maintenance. Soaps and detergents are organic compounds that remove grease and dirt through chemical interactions with fats. Many cosmetics, shampoos, perfumes, and disinfectants also contain organic molecules because they are effective, biodegradable, and compatible with human skin.
Organic Alcohols in Daily Life
Alcohols such as ethanol are widely used as disinfectants, solvents, fuels, and preservatives. Ethanol is commonly found in hand sanitizers, medicines, perfumes, and alcoholic beverages, making it one of the most frequently used organic compounds in everyday life.
4. Organic Compounds as Fuels and Energy Sources
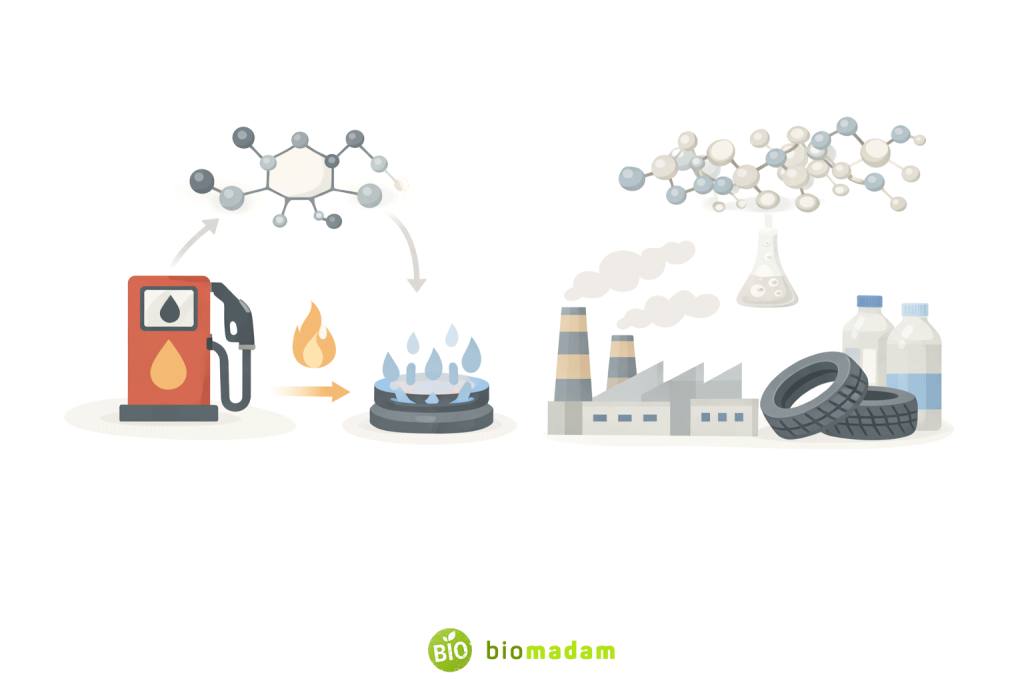
Hydrocarbons
Hydrocarbons contain only carbon and hydrogen and are major energy sources.
Common examples:
- Methane (natural gas)
- Propane and butane (LPG)
- Ethane and ethyne (industrial fuel)
Uses:
- Cooking fuel
- Vehicle fuel
- Electricity generation
- Heating
Hydrocarbons store large amounts of energy within their carbon–hydrogen bonds. When these bonds break during combustion, energy is released in the form of heat and light. This property explains why hydrocarbons have been widely used as fuels for cooking, transportation, and electricity generation throughout human history.
5. Organic Compounds in Industry
Organic compounds are essential raw materials for:
- Plastics and polymers
- Detergents and soaps
- Dyes, inks, and perfumes
- Rubber and synthetic fibers
- Fermentation and pharmaceutical industries
Starch, for example, is biodegradable and used in eco-friendly industrial applications.
The versatility of organic compounds in industry arises from carbon’s ability to form long chains, branched structures, and rings. These structural variations allow manufacturers to design materials with specific properties, such as flexibility, strength, biodegradability, or resistance to heat and chemicals.
For example, polyethylene and PVC are used in plastic manufacturing, sodium stearate is used in soap production, synthetic dyes are derived from aromatic organic compounds, and rubber polymers are widely used in tires and industrial materials.
Which Is the Most Abundant Organic Compound?
Among organic compounds, carbohydrates are the most abundant and widely used. They occur extensively in plants, animals, and food products and serve as the primary source of energy in living organisms.
Examples include:
- Glucose
- Sucrose
- Starch
- Cellulose
Carbohydrates are abundant not only in plants, where they are produced through photosynthesis, but also in human diets and metabolic pathways. They serve as the primary energy source for living organisms and form structural components such as cellulose in plant cell walls, making them the most widespread organic compounds in nature.
Importance of Organic Compounds
- Essential for life processes
- Major source of energy
- Basis of medicines and healthcare
- Support industrial and agricultural development
- Integral to food, fuel, and consumer products
Organic compounds form the chemical foundation of life and modern civilization. Their broader significance is explained further in the Importance of Organic Compound. From providing energy and building biological structures to enabling medicines, fuels, and industrial materials, their versatility makes them indispensable. Without organic compounds, neither living systems nor technological development would be possible.
Conclusion
Organic compounds play a vital role in everyday life by supporting nutrition, health, energy production, and industrial growth. From carbohydrates and fats in food to hydrocarbons used as fuels and organic acids in medicine, these compounds are indispensable for modern living. Understanding their uses helps students connect chemistry concepts with real-world applications, as explained in the Importance of Chemistry in Daily Life.
FAQs
What are organic compounds?
Organic compounds are carbon-based chemical substances, mainly containing carbon and hydrogen.
Why are organic compounds important in daily life?
They provide food, fuel, medicines, and materials used in everyday products.
Are all carbon compounds organic?
No. Carbonates, cyanides, and carbides are inorganic compounds.
Which organic compound is most abundant?
Carbohydrates are the most abundant organic compounds.
Are medicines organic compounds?
Yes, most drugs and pharmaceuticals are organic compounds.

The Science Editorial Team creates clear, accurate, and student-friendly explanations of scientific concepts across biology, chemistry, physics, taxonomy, anatomy, and related life sciences. Content is written for educational and informational purposes using standard academic references and reliable sources. Read more about the → Science Editorial Team
